Interaction of arylglyoxals with thioureas
DOI:
https://doi.org/10.15421/081411Keywords:
2-thiohydantoine, arylglyoxals, thiazolidinoneAbstract
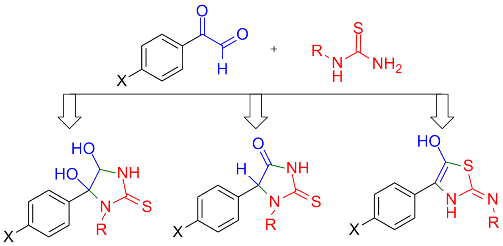
Literature data about the reaction of arylglyoxals with thiourea has been discussed. It has been shown that no systematic studies in this issue had ever been carried out, furthermore some data is contradictory. The reaction of arylglyoxals with thiourea is reported to be a stepwise process that depends on at least two factors. It is reaction conditions such as temperature and solvent. Also the nature of glyoxal may have crucial influence on the structure of products. In this article has been provided the convincing evidence of formation three different kinds of heterocyclic systems upon reaction of p-substituted phenylglyoxals with N-methyl and N-phenylthiourea.References
Schubert, M. P. Combination of thiol acids with methylglyoxal. J. Biol. Chem., 1935, Vol. 111, p. 671–678.
McKay, W. R., Proctor, G. R. Removal of toluene-p-sulphonyl groups from sulphonamides. Part 5. Reactions of phenylglyoxal imines and some tosylimines. J. Chem. Soc., Perkin Trans. 1, 1981, p. 2443–2450.
Paul, S., Gupta, M., Gupta, R., Loupy, A. Microwave Assisted Synthesis of 1,5-Disubstituted Hydantoins and Thiohydantoins in Solvent-Free Conditions. Synthesis, 2002, p. 75–78.
Ettari, R., Pinto, A., Micale, N. Synthesis and anti-HIV activity evaluation of new phenyl ethyl thiourea (PET) derivatives. ARCIVOC, 2009, p. 227–234.
Anishchenko, A. A., Shtamburg, V. G., Volosyuk, V. V., Olefir, D. A., Mazepa, A. V. Condensation of p-chlorophenylglyoxal with phenylthiourea. Visn. Dnipropetr. Univ.: Khim., 2012, vol. 20, no. 18, p. 110–112. [in Russian]
Downloads
Published
Issue
Section
License
Copyright (c) 2014 Oles Honchar Dnipropetrovsk National University

This work is licensed under a Creative Commons Attribution 4.0 International License.
- Authors reserve the right of attribution for the submitted manuscript, while transferring to the Journal the right to publish the article under the Creative Commons Attribution License. This license allows free distribution of the published work under the condition of proper attribution of the original authors and the initial publication source (i.e. the Journal)
- Authors have the right to enter into separate agreements for additional non-exclusive distribution of the work in the form it was published in the Journal (such as publishing the article on the institutional website or as a part of a monograph), provided the original publication in this Journal is properly referenced
- The Journal allows and encourages online publication of the manuscripts (such as on personal web pages), even when such a manuscript is still under editorial consideration, since it allows for a productive scientific discussion and better citation dynamics (see The Effect of Open Access).

