New method of oligomeric acetoxymethylsiloxanes synthesis
DOI:
https://doi.org/10.15421/081422Keywords:
oligomer acetoxymethylsiloxanes, synthesis, structure, intermediate products, α-alcohols, polysiloxaneurethanesAbstract
The studies proposed a new method of synthesis of various structures oligomer acetoxymethylsiloxanes with the use of heterofunctional condensation reaction between acetoxymethyldimethylacetoxysilane and alkali metals siloxanolats. The indicated oligomer used as intermediate products in the synthesis of different compounds, for example, organosilicon carbofunctional α-alcohols, which are the starting point for production of polysiloxaneurethanes and other polymeric and composite materials with a number of useful properties. It has been found that the reaction proceeds in high yield (over 91%) in the temperature range 0–110°С in inert (under synthesis conditions) solvents – toluene or dioxane. Reaction proceed in heterogeneous or homogeneous conditions depending on the structure of siloxanolats. Take into account that typical segregation acetoxymethyl group with creation of methylacetate which proceeds under influence of nucleophilic agent – alkali metal siloxanolat. Required conditions for successful reaction heterofunctional condensation is the absence in a reaction of mass proton-donating agents which cause passing of concurrent reactions (water, alcohols, acids etc.).
References
Kuznetsova, V. P., Laskovenko, N. N., Zapunnaja, K. V. Organosilicon polyurethanes. Kyiv: Naukova dumka, 1984, 224 p. [in Russian]
Lai Y.–Ch., Quinn E. T. US Patent no. 7132492, 2006.
Lai Y.–Ch., Quinn E. T. US Patent no. 7132493, 2006.
Sukachev, V. V., Buria, A. I. Polysiloxane urethane coating based on the organosilicon carbofunctional polyatomic α alcohols. Visnyk Skhidnoukr. nats. un-tu im. V. Dalya, 2010, no. 10 (152), p. 180–184. [in Russian]
Sukachev, V. V., Buria, A. I., Ebitsh, Y. R. Elastomers modification with silicon–urethans. 11th International conference, Plavya, Ukraine, 2011, p. 27–30, http://www.conference.kiev.ua/?page_id=356. [in Russian]
Sukachev, V. V., Tchigvintseva, O. P. Studing the process of hydrolytic co-condensation organoacetoxysilanes. Synthe-sis acetoxymethylsiloxanes. Visnyk nats. teсhn. in-tu «KHPI»: zb. nauk. pr., 2013, no. 64, p. 148–156. [in Russian]
Krechkov, A. P., Bork, V. A., Bondarevskaia, E. A., Muchliaeva, L. V., Siavtsillo, S. V., Chemiatenkova, V. T. Practical guidance on the analysis of monomeric and polymeric organosilicon compounds. Moskow: Goskhimizdat, 1962, 544 p. [in Russian]
Smith, R. C., Kellum, G. E. Rapid condensation procedure for determination of hydroxyl in silicone materials. Anal. Chem., 1967, vol. 39, no.3, p. 338–340.
Voronkov, M. G., Maletina, E. A., Roman, V. K. Heterosiloxanes. Novosibirsk: Nauka, 1984, 272 p. [in Russian]
Voronkov, M. G., Milechkevitsh, V. P., Jugelevskii, Ju. A. Siloxane bond. Novosibirsk: Nauka, 1976, 416 p. [in Russian]
Bostick, E. E., Fessler, W. A. US Patent no. 3578726, 1971.
Sobolevskaia, M. V., Tchistiakova, L. A., Nazarova, D. V., Kirillina, V. V. Synthesis of α, ω-hexaorganopolydimethyl-polymethylphenylsiloxanes regularly alternating with dimethyl- and methylphenylsiloxy units in the chain. Plasticheskiye massy, 1962, no. 10, p. 17–21. [in Russian]
Bellami, L. J. The infra-red spectra of complex molecules. Moskow: Izdat. inostran. lit, 1963, 590 p. [in Russian]
Bellami, L. J. Advances in infrared group frequencies. Moskow: Mir, 1971, 320 p. [in Russian]
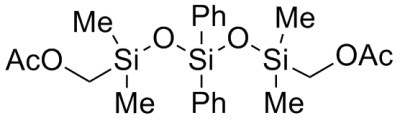
Downloads
Published
Issue
Section
License
Copyright (c) 2014 Oles Honchar Dnipropetrovsk National University

This work is licensed under a Creative Commons Attribution 4.0 International License.
- Authors reserve the right of attribution for the submitted manuscript, while transferring to the Journal the right to publish the article under the Creative Commons Attribution License. This license allows free distribution of the published work under the condition of proper attribution of the original authors and the initial publication source (i.e. the Journal)
- Authors have the right to enter into separate agreements for additional non-exclusive distribution of the work in the form it was published in the Journal (such as publishing the article on the institutional website or as a part of a monograph), provided the original publication in this Journal is properly referenced
- The Journal allows and encourages online publication of the manuscripts (such as on personal web pages), even when such a manuscript is still under editorial consideration, since it allows for a productive scientific discussion and better citation dynamics (see The Effect of Open Access).

