CHEMICAL AND PHYSICO-CHEMICAL FEATURES OF MODIFIED BIOCARBON ECOSORBENT FOR BINDING HEAVY METAL IONS
DOI:
https://doi.org/10.15421/jchemtech.v32i2.298468Keywords:
adsorption, cadmium, lead, zinc, copper, bagasse, rice husk, pyrolysis, biochar, chemical modification, sulfate-reducing bacteria, sulfides.Abstract
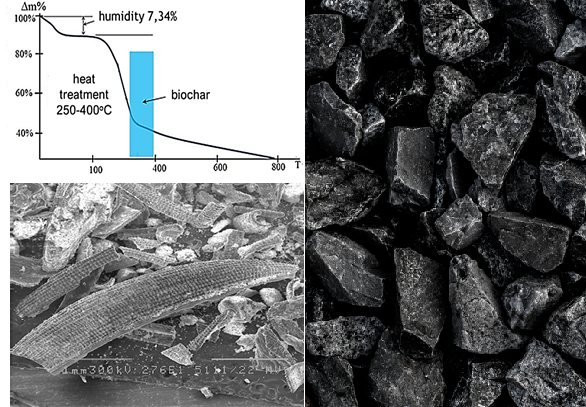
The possibility of obtaining ecosorbents based on biologically and chemically modified carbon material from vegetable raw materials for the removal of Cd (II), Cu (II), Pb (II) and Zn (II) ions from aqueous solutions was studied. The peculiarities of obtaining an ecosorbent from sugar cane bagasse and rice husk are substantiated. Sulfur-containing biocarbon ecosorbent is obtained by one-stage pyrolysis of raw materials together with sulfur-containing reagents at a temperature of 350–400 °C. The temperature parameters and the duration of the pyrolysis process were determined experimentally from the conditions of the maximum content of biochar in the pyrolyzate. Sulfur-containing biochar has a high absorption capacity for heavy metal ions (more than 80–90 %) compared to the original biochar. Its specificity is due to the formation of insoluble metal sulfides on the surface and in the pores of the sorbent. Microbiological modification leads to the creation of biosorption material for strong binding of heavy metal ions. Sulfate-reducing bacteria immobilized on the surface of biochar are also able to convert sorbed heavy metals into insoluble sulfide forms. A comprehensive study of the structural-porous and sorption properties of the original and modified biochar was conducted. It was established that chemical and microbiological modification of biochar increases its sorption capacity due to the improvement of structural-porous and ion-exchange properties of the material. But the key property of ecosorbents from pyrolyzed and modified vegetable raw materials is the ability to form insoluble sulfide forms of metals on the surface and in the pores of the ecosorbent.
References
Chaffai, R., Koyama, H. (2011). Heavy metal tolerance in Arabidopsis thaliana, Adv. Bot. Res, 60, 1–49. doi: 10.1016/B978-0-12-385851-1.00001-9.
Meyer, S.; Glaser, B., Quicker, P. (2011). Technical, economical, and climate-related aspects of biochar production technologies. A literature review, Environ. Sci. Technol., 45, 9473–9483. doi: 10.1021/es201792c
Reddy, D.H.K., Seshaia, K., Reddy, A.V.R., Rao, M.M.
Wang, M.C. (2010). Biosorption of Pb2+ from Aqueous Solutions by Moringa oleifera Bark: Equilibrium and Kinetic Studies, Journal of Hazardous Materials, 174, 831–838.
Ngah, W.S.W., Hanafiah, M.A.K.M. (2008). Removal of Heavy Metal Ions from Wastewater by Chemically Modi-fied Plant, Wastes as Adsorbents: A Review, Bioresource Technology, 99, 3935–3948. http://dx.doi.org/10.1016/j.biortech.2007.06.011
Tarnovsky, D.V., Tsyba, M.M., Kuznetsova, L.S., Khodakovska, T.A., Romanova, I.V. (2021). Physico-chemical properties of cerium and ferric doped titanium hydroxides synthesized by two methods, Journal of Chemystry and Technologies, 29(2), 192–199. https://doi.org/10.15421/jchemtech.v29i2.232199 .
Atafar, Z., Mesdaghinia, A., Nouri, J., Homaee, M., Yunesian, M., Ahmadimoghaddam, M., Mahvi, A. H. (2010). Effect of fertilizer application on soil heavy metal concentration, Environ. Monit, Asses., 160, 83–89. doi: 10.1007/s10661-008-0659-x
Liu, X., Na, S., Lu, Z. D., Liu, Z. (2013). Adsorption, Concentration, and Recovery of Aqueous Heavy Metal Ions with the Root Powder of Eichhornia crassipes, Ecological Engineering, 60, 160–166. http://dx.doi.org/10.1016/j.ecoleng.2013.07.039
Boonstra J., Dijman Н., Lawrence R. And Buisman C.J.N. (2002). Water treatment and metals recycling using biogenic sulfide. Recycling and waste treatment in mineraland metal processing: technical and economic aspects, Lulea, Sweden., l, 691-698.
Xu, M., Yin, P., Liu, X., Tang, Q., Qu, R., Xu, Q. (2013). Utilization of Rice Husks Modified by Organomultipho-sphonic Acids as Low-Cost Biosorbents for Enhanced Adsorption of Heavy Metal Ions, Bioresource Technology, 149. 420–424. http://dx.doi.org/10.1016/j.biortech.2013.09.075
Kwon, J.S., Yun, J.H., Kim, S.O., Jo, H.Y. (2010). Removal of Divalent Heavy Metals (Cd, Cu, Pb, and Zn) and Arsenic (III) from Aqueous Solutions Using Scoria: Kinetics and Equilibria of Sorption, Journal of Hazardous Materials, 174, 307–313. http://dx.doi.org/10.1016/j.jhazmat.2009.09.052
M N Mohamad Ibrahim , W S Wan Ngah, M S Norliyana, W R Wan Daud, M Rafatullah, O Sulaiman, R Hashim (2010). A Novel Agricultural Waste Adsorbent for the Removal of Lead (II) Ions from Aqueous Solutions, Journal of Hazardous Mate-rials, 182, 377–385. http://dx.doi.org/10.1016/j.jhazmat.2010.06.044
Ding, D., Zhou, L., Kang, F., Yang, S., Chen, R., Cai, T., Duan, X., Wang, S. (2020). Synergistic Adsorption and Oxidation of Ciprofloxacin by Biochar Derived from Metal-Enriched Phytoremediation Plants: Experimental and Computational Insights, ACS Applied Materials & Interfaces, 12, 48, 53788–53798.
Demirbas, A. (2008). Heavy Metal Adsorption onto Agro-Based Waste Materials: A Review, Journal of Hazardous Materials, 157, 220–229. http://dx.doi.org/10.1016/j.jhazmat.2008.01.024
Kaur, R., Singh, J., Khare, R., Cameotra, S.S., Ali, A. (2013). Batch Sorption Dynamics, Kinetics and Equilibrium Studies of Cr(VI), Ni(II) and Cu(II) from Aqueous Phase Using Agricultural Residues, Applied Water Science, 3, 207–218
dos Reis, G. S., Bergna, D., Grimm, A., Lima, E. C., Hu, T., Naushad, M., Lassi, U. (2023). Preparation of highly porous nitrogen-doped biochar derived from birch tree wastes with superior dye removal performance. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 669,131–493.
https://doi.org/10.1016/j.colsurfa.2023.131493
Liang, S., Guo, X., Tian, Q. (2011). Adsorption of Pb2+ and Zn2+ from Aqueous Solution by Sulfured Orange Peel, Desalination, 275, 212–216. http://dx.doi.org/10.1016/j.desal.2011.03.001
Kamar, F.H., Nechifor, A.C., Mohammed, A.A., Albu, P.C., Craciun, M.E. (2015). Removal of lead and cadmium ions from aqueous solutions using walnut shells as low-cost adsorbent materials, Revista de Chimie, 66(5), 615–620.
Ding, D., Zhou, L., Kang, F., Yang, Sh., Chen, R., Cai, T., Duan, X., Wang, Sh. (2020). Synergistic Adsorption and Oxidation of Ciprofloxacin by Biochar Derived from Metal-Enriched Phytoremediation Plants: Experimental and Computational Insights, ACS Applied Materials & Interfaces, 12(48), 53788–53798.
Sungsinchai, S., Niamnuy, Ch., Devahastin, S., Chen, X. D., Chareonpanich, M. (2023). Effect of the Structure of Highly Porous Silica Extracted from Sugarcane Bagasse Fly Ash on Aflatoxin B1 Adsorption. ACS Omega, 8(22), 19320–19328. https://doi.org/10.1021/acsomega.2c08299
Hamed, R., Jodeh, S., Hanbali, G., Safi, Z., Berisha, A., Xhaxhiu, K., Dagdag, O. (2022). Eco-Friendly Synthesis and Characterizationof Double-Crossed Link 3D Graphene Oxide Functionalized with Chitosan for Adsorption of Sulfamethazine from Aqueous Solution: Experimental and DFT Calculations. Front. Environ. Sci., 10, 930693.
El-Moselhy, K.M., Azzem, M.A., Amer, A., Al-Prol, A.E. (2017). Adsorption of Cu (II) and Cd (II) from aqueous solution by using rice husk adsorbent, Physical Chemistry: An Indian Journal, 12(2), 1–13.
Downloads
Published
Issue
Section
License
Copyright (c) 2024 Oles Honchar Dnipro National University

This work is licensed under a Creative Commons Attribution 4.0 International License.
- Authors reserve the right of attribution for the submitted manuscript, while transferring to the Journal the right to publish the article under the Creative Commons Attribution License. This license allows free distribution of the published work under the condition of proper attribution of the original authors and the initial publication source (i.e. the Journal)
- Authors have the right to enter into separate agreements for additional non-exclusive distribution of the work in the form it was published in the Journal (such as publishing the article on the institutional website or as a part of a monograph), provided the original publication in this Journal is properly referenced
- The Journal allows and encourages online publication of the manuscripts (such as on personal web pages), even when such a manuscript is still under editorial consideration, since it allows for a productive scientific discussion and better citation dynamics (see The Effect of Open Access).

