HPLC METHOD VALIDATION AND UNCERTAINTY DETERMINATION FOR QUANTIFICATION OF KRESOXIM METHYL FUNGICIDE IN CHILI
DOI:
https://doi.org/10.15421/jchemtech.v34i1.328444Keywords:
Kresoxim methyl; method validation; HPLC; measurement uncertaintyAbstract
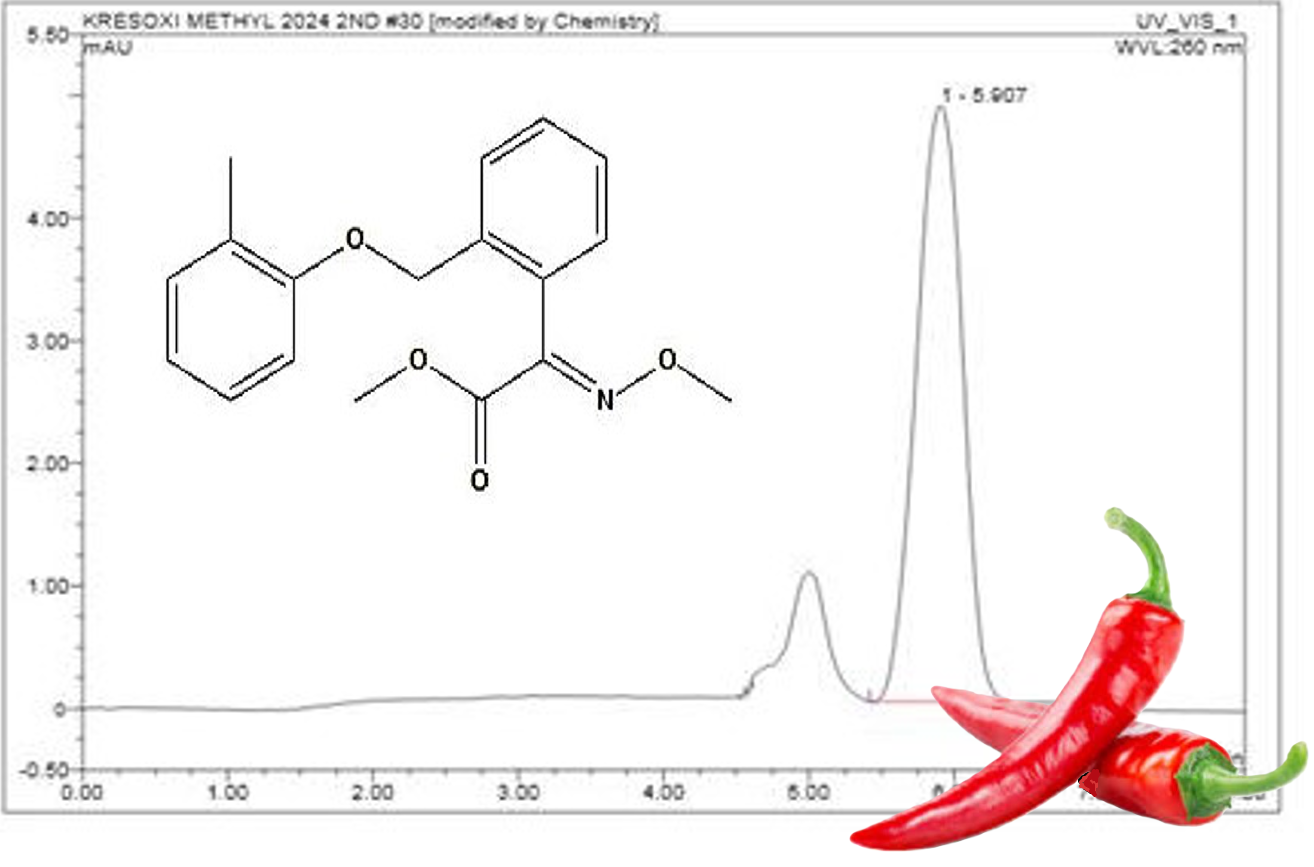
A simple and efficient multi-residue analytical method was developed and validated for the determination of Kresoxim methyl (KM) fungicide applied on chili crop for controlling of several fungal diseases. The fungicide residues from chili samples were extracted using modified QuEChERS method, followed by analysis through HPLC–UV. Method validation parameters viz. specificity, linearity, matrix effects, LOD, LOQ, recovery, accuracy and precision, robustness and estimation of measurement uncertainty were evaluated. Specificity of the method based on the chromatographic peak purity was observed in the chromatograms of KM, and calibration curve was found to be linear with R2> 0.99. Matrix effect for KM in chili was < ±20 %. The recovery studies were conducted by spiking the samples at three (LOQ, 5LOQ and 10LOQ) concentration levels. The average recovery of KM at all the three concentrations ranged between 84–86 % with percent RSD < 3 %. Measurement uncertainty (MU) can be helpful while deciding the compliance of chili samples against the established MRL. Hence both type A and type B uncertainties were considered for calculation of combined uncertainty (Uc) in the method validation process. The combined uncertainty (Uc) was below 25 % default value which is normally considered satisfactory for nonfatty matrixes (fruit, vegetables, and grain) by many regulatory authorities for enforcement decisions. The proposed method validation for KM in chili was simple, rapid, and cost effective with high accuracy and sensitivity requiring minimum use of organic solvents.
References
Sauter, H., Steglich, W., &Anke, T. (1999). Strobilurins: Evolution of a new class of active substances. AngewandteChemie International Edition, 38(10),1328–1349. https://doi.org/10.1002/(SICI)1521-3773(19990517)38:10<1328::AID-ANIE1328>3.0.CO;2-1
Herms, S., Seehaus, K., Koehle, H., &Conrath, U. (2002). A strobilurin fungicide enhances the resistance of tobacco against tobacco mosaic virus and Pseudomonas syringaepvtabaci. Plant Physiology, 130(1), 120–127. https://doi.org/10.1104/pp.004432
Bartlett, D. W., Clough, J. M., Godwin, J. R., Hall, A. A., Hamer, M., & Parr-Dobrzanski, B. (2002). The strobilurin fungicides. Pest Management Science, 58(7), 649–662. https://doi.org/10.1002/ps.520
Valavanidis, A. (2016). Pesticide residues in fruit, vegetables and food. How dangerous are to human health? Studies of pesticide residues in food in European countries and in Greece, and risk to consumer’s health [Doctoral dissertation, University of Athens].
Carvalho, F. P. (2017). Pesticides, environment, and food safety. Food and Energy Security, 6(2), 48–60. https://doi.org/10.1002/fes3.108
Hussain, F., Shaukat, S. S., Muhammad, A., Farzana, U., & Muhammad, A. (2013). Filamentous fungi infecting fruits and leaves of Capsicum annum L. in lower Sindh. International Journal of Biology and Biotechnology, 10(1), 109–116.
Gul, S., Khanum, K., & Mujtaba, N. (2015). New validated method for analysis of silymarin in polyherbal formulation (aqueous extract, oral liquid and solid dosage). Chemistry International, 1(3), 103–106.
Ramsey, M. H., & Ellison, S. L. R. (Eds.). (2007). Eurachem/EUROLAB/CITAC/Nordtest/AMC Guide: Measurement uncertainty arising from sampling: A guide to methods and approaches. Eurachem.
Apparao, K., Surendra Babu, M. S., Rao, T. N., Patrudu, T. B., &Basaveswara Rao, M. V. (2016). Determination of kresoxim-methyl residues in different types of Indian tropical soils. Journal of Soil Science Research, 1(1), 1–6.
Rahman, M. M., Park, J. H., Abd El-Aty, A. M., Choi, J. H., Cho, S. K., Yang, A., Park, K. H., & Shim, J. H. (2013). Analysis of kresoxim-methyl and its thermolabile metabolites in Korean plum: An application of pepper leaf matrix as a protectant for GC amenable metabolites. Journal of Separation Science, 36(1), 203–211. https://doi.org/10.1002/jssc.20120061
Carlos, A. T., Toloda, J., Almeida, M. S., Silva, L. O. P., Macedo, R. C., Lamounier, A. P., Aucelio, R. Q., & da Cunha, A. L. M. C. (2020). Determination of kresoxim-methyl and its main metabolite in natural waters by HPLC-DAD and fluorescence. Analytical Letters, 53(14), 2202–2221. https://doi.org/10.1080/00032719.2020.1733589
Majumder, S., Verma, K. C., Rani, V., Rani, A. T., Pandey, K. K., & Singh, J. (2022). Residue dynamics and food safety evaluation of fungicide kresoxim-methyl in green chilli (Capsicum annum L.). International Journal of Environmental Analytical Chemistry, 102(19), 7433–7443. https://doi.org/10.1080/03067319.2020.1830986
Srivastava, A., Singh, G., & Srivastava, P. C. (2021). Method validation for determination of nine pesticides in okra and their mitigation using different solutions. PLoS ONE, 16(12), e0260851. https://doi.org/10.1371/journal.pone.0260851
Bakanov, N., Honert, C., Eichler, L., Lehmann, G. U., Schulz, R., & Brühl, C. A. (2023). A new sample preparation approach for the analysis of 98 current-use pesticides in soil and herbaceous vegetation using HPLC-MS/MS in combination with an acetonitrile-based extraction. Chemosphere, 331, 138840. https://doi.org/10.1016/j.chemosphere.2023.138840
Lehotay, S. J. (2011). QuEChERS sample preparation approach for mass spectrometric analysis of pesticide residues in foods. In Mass Spectrometry in Food Safety. Humana Press. https://doi.org/10.1007/978-1-61779-136-9_4
European Commission. (2021). Analytical quality control and method validation procedures for pesticide residues analysis in food and feed (Document No. SANTE/11312/2021). European Commission.
Naz, S., Vallejo, M., García, A., &Barbas, C. (2014). Method validation strategies involved in non-targeted metabolomics. Journal of Chromatography A, 1353, 99–105. https://doi.org/10.1016/j.chroma.2014.04.071
Vander Heyden, Y., Nijhuis, A., Smeyers-Verbeke, J., Vandeginste, B. G. M., &Massart, D. L. (2001). Guidance for robustness/ruggedness tests in method validation. Journal of Pharmaceutical and Biomedical Analysis, 24(5-6), 723–753. https://doi.org/10.1016/S0731-7085(00)00529-X
Yar, A., Ansari, T. M., Raza, A., Javeed, Z., & Asif, M. (2023). Development and validation of novel HPLC method for determination of nitenpyram insecticide in commercial samples. Journal of Xi'an Shiyou University, Natural Science Edition, 19(7), 810–822.
Sanyal, D., Rani, A., Alam, S., Gujral, S., & Gupta, R. (2011). Development, validation, and uncertainty measurement of multi-residue analysis of organochlorine and organophosphorus pesticides using pressurized liquid extraction and dispersive-SPE techniques. Environmental Monitoring and Assessment, 182, 97–113. https://doi.org/10.1007/s10661-010-1861-1
Eurachem/CITAC. (2000). Quantifying uncertainty in analytical measurement (2nd ed., Guide CG 4). http://www.measurementuncertainty.org
Alder, L., Korth, W., Patey, A. L., van der Schee, H. A., &Schoeneweiss, S. (2001). Estimation of measurement uncertainty in pesticide residue analysis. Journal of AOAC International, 84(5), 1569–1578. https://doi.org/10.1093/jaoac/84.5.1569
Downloads
Published
Issue
Section
License
Copyright (c) 2026 Oles Honchar Dnipro National University

This work is licensed under a Creative Commons Attribution 4.0 International License.
- Authors reserve the right of attribution for the submitted manuscript, while transferring to the Journal the right to publish the article under the Creative Commons Attribution License. This license allows free distribution of the published work under the condition of proper attribution of the original authors and the initial publication source (i.e. the Journal)
- Authors have the right to enter into separate agreements for additional non-exclusive distribution of the work in the form it was published in the Journal (such as publishing the article on the institutional website or as a part of a monograph), provided the original publication in this Journal is properly referenced
- The Journal allows and encourages online publication of the manuscripts (such as on personal web pages), even when such a manuscript is still under editorial consideration, since it allows for a productive scientific discussion and better citation dynamics (see The Effect of Open Access).

