EFFECT Of OH-/M2+ RATIO ON THE PROPERTIES OF Fe/CoFe2O4 COMPOSITES OBTAINED BY HYDROTHERMAL METHOD
DOI:
https://doi.org/10.15421/jchemtech.v34i1.332246Keywords:
magnetic properties, spinel, X-ray phase analysisAbstract
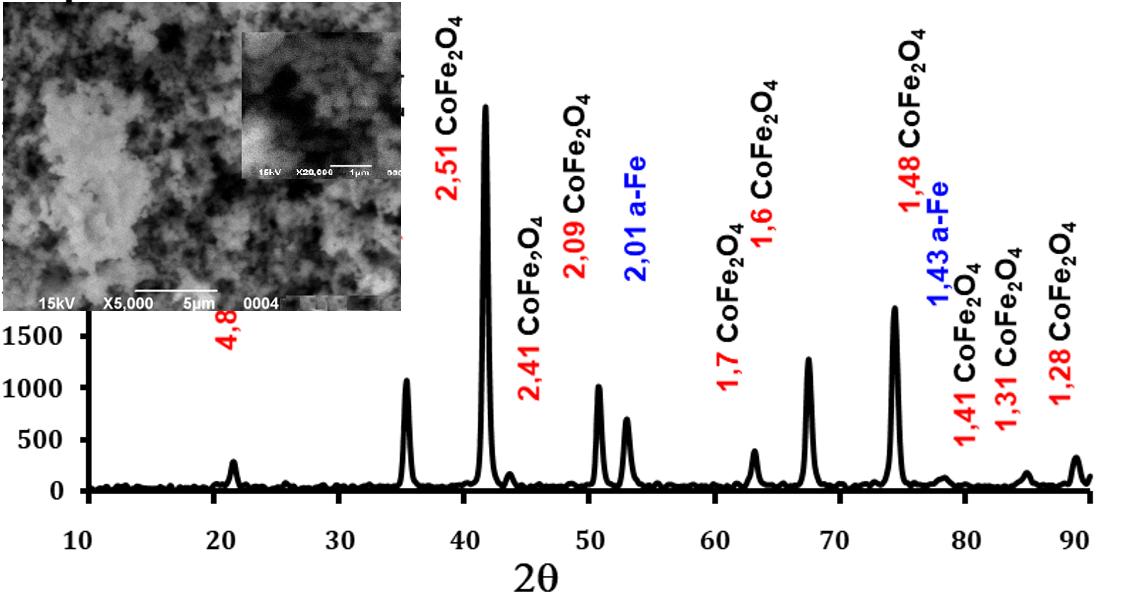
Nanodisperse composites were synthesized by hydrothermal method at elevated pressure. To study the influence of OH-/M2+ concentration ratio on phase composition, degree of crystallinity, average crystallite size, magnetic and absorption properties of Fe/CoFe2O4 composites, a series of Fe/CoFe2O4 composites was obtained by varying synthesis conditions. The obtained composites were characterized by X-ray diffraction, scanning electron microscopy, vibrational magnetometry and microwave analysis. A larger excess of precipitant leads to a higher degree of crystallinity and a larger average crystallite size of CoFe2O4. With an increase in the [OH– /Me2+] ratio, the diffraction peaks significantly increase in sharpness and intensity, indicating the formation of crystals with higher crystallinity and average crystallite size. Noticeable shifts in the positions of the diffraction peaks are also found for all samples, while the value of the crystal lattice constant decreases (8.3901–8.3699 Å). Studies using vibrational magnetometry have shown that the dependence of the saturation magnetization (Ms) correlates with the a-Fe content. The maximum values of the saturation magnetization of 200 Emu/g and the coercive force of 900 Oe correspond to a ratio of OH-/M2+ concentrations equal to 4.6. All obtained samples effectively absorb electromagnetic radiation in the range of 8–10 GHz.
References
Arora, K., Ledwani, L., & Komal, Dr. (2025). A Comprehensive Review on the Synthesis and Therapeutic Potential of Cobalt Ferrite (CoFe2O4) Nanoparticles. Chemistry Select, 10(1), e202404136. https://doi.org/10.1002/slct.202404136
Chamaraja, N. A., Swamy, N. K., Shivakumar, V., Kavya, H. V., & Bvaraju, M. (2025). Evaluation of peroxidase mimetic properties of cobalt ferrite (CoFe2O4) nanoparticles and their applications in the assay of aminophenols in water samples. Nanotechnology for Environmental Engineering, 10(1), 15. doi:10.1007/s41204-025-00413-z
Patil, P. S., Bhagat, S., Kokate, R., & Jadhav, K. M. (2025). Investigations of structural, optical and magnetic properties of CoFe2O4 nano-catalyst for hydrogen production applications. In AIP Conference Proceedings, 3198(1), 020032. https://doi.org/10.1063/5.0248275
Jasrotia, R., Prakash, J., Saddeek, Y. B., Alluhayb, A. H., Younis, A. M., Lakshmaiya, N., Prakash, C., Aly K.A., Sillanpa M., Ismail, Y., Kandwal, A., & Sharma, P. (2025). Cobalt ferrites: Structural insights with potential applications in magnetics, dielectrics, and Catalysis. Coordination Chemistry Reviews, 522, 216198. doi:10.1016/j.ccr.2024.216198
Ismail, M., Ali, N. A., Sazelee, N. A., Muhamad, S. U., Suwarno, S., & Idris, N. H. (2022). CoFe2O4 synthesized via a solvothermal method for improved dehydrogenation of NaAlH4. International Journal of Hydrogen Energy, 47(97), 41320–41328. https://doi.org/10.1016/j.ijhydene.2022.01.215
Bayça, F. (2024). Characterization and magnetic properties of CoFe2O4 nanoparticles synthesized by the co‐precipitation method. International Journal of Applied Ceramic Technology, 21(1), 544-554. https://doi.org/10.1111/ijac.14518
Gingasu, D., Culita, D. C., Calderon Moreno, J. M., Marinescu, G., Bartha, C., Oprea, O., Preda, S., Chifiriuc, M. C., & Popa, M. (2023). Synthesis of CoFe2O4 through wet ferritization method using an aqueous extract of eucalyptus leaves. Coatings, 13(7), 1250. https://doi.org/10.3390/coatings13071250
Heydaryan, K., Kashi, M. A., & Montazer, A. H. (2022). Tuning specific loss power of CoFe2O4 nanoparticles by changing surfactant concentration in a combined co-precipitation and thermal decomposition method. Ceramics International, 48(12), 16967–16976. doi:10.1016/j.ceramint.2022.02.251
Vishwas, M., Babu, K. V., Gowda, K. A., & Gandla, S. B. (2022). Synthesis, characterization and photo-catalytic activity of magnetic CoFe2O4 nanoparticles prepared by temperature controlled co-precipitation method. Materials Today: Proceedings, 68(11), 497–501. doi:10.1016/j.matpr.2022.07.429
Boussafel, H., Sedrati, C., & Alleg, S. (2024). Green synthesis of NiFe2O4, CoFe2O4, and NiО. 5CoО. 5Fe2O4 by sol–gel autocombustion method using olive leaf extract as fuel. Applied Physics A, 130(6), 374. doi:10.1007/s00339-024-07547-y
Shakirzyanov, R. I., Kozlovskiy, A. L., Zdorovets, M. V., Zheludkevich, A. L., Shlimas, D. I., Abmiotka, N. V., Kazantsev, P.A., Zubar, T.I., Trukhanov, S. V., & Trukhanov, A. V. (2023). Impact of thermobaric conditions on phase content, magnetic and electrical properties of the CoFe2O4 ceramics. Journal of Alloys and Compounds, 954, 170083. https://doi.org/10.1016/j.jallcom.2023.170083
Huang, T., Qiu, Z., Hu, Z., & Lu, X. (2021). Novel method of preparing hierarchical porous CoFe2O4 by the citric acid-assisted sol-gel auto-combustion for supercapacitors. Journal of Energy Storage, 35, 102286. https://doi.org/10.1016/j.est.2021.102286
Ravindra, A. V., & Ju, S. (2023). Mesoporous CoFe2O4 nanocrystals: Rapid microwave-hydrothermal synthesis and effect of synthesis temperature on properties. Materials Chemistry and Physics, 303, 127818. https://doi.org/10.1016/j.matchemphys.2023.127818
Prasad, K., Sreekanth, T. V. M., Yoo, K., & Kim, J. (2023). Surfactant-assisted hydrothermal synthesis of CoFe2O4 nanostructures and their application in the oxygen evolution reaction. Materials Letters, 349, 134859. doi:10.1016/j.matlet.2023.134859
Xiangfeng, C., Dongli, J., Yu, G., & Chenmou, Z. (2006). Ethanol gas sensor based on CoFe2O4 nano-crystallines prepared by hydrothermal method. Sensors and Actuators B: Chemical, 120(1), 177–181. doi:10.1016/j.snb.2006.02.008
Zhao, D., Wu, X., Guan, H., & Han, E. (2007). Study on supercritical hydrothermal synthesis of CoFe2O4 nanoparticles. The Journal of supercritical fluids, 42(2), 226–233. doi:10.1016/j.supflu.2007.03.004
Ansari, S. M., Younis, A., Kolekar, Y. D., & Ramana, C. V. (2025). Cobalt ferrite nanoparticles: The physics, synthesis, properties, and applications. Applied Physics Reviews, 12(2), 021308. https://doi.org/10.1063/5.0244555
Tatarchuk, T., Shyichuk, A., Kotsyubynsky, V., & Danyliuk, N. (2025). Catalytically active cobalt ferrites synthesized using plant extracts: Insights into structural, optical, and catalytic properties. Ceramics International, 51(4), 4988–4999. https://doi.org/10.1016/j.ceramint.2024.11.470
Yadav, S. M., Kumar, S., Kumar, M., Ghosh, A., & Saini, D. S. (2024). High-temperature variable range hopping conduction and dielectric relaxation in CoFe2O4 ceramic. Open Ceramics, 17, 100517. https://doi.org/10.1016/j.oceram.2023.100517
Varma, P. R., Manna, R. S., Banerjee, D., Varma, M. R., Suresh, K. G., & Nigam, A. K. (2008). Magnetic properties of CoFe2O4 synthesized by solid state, citrate precursor and polymerized complex methods: A comparative study. Journal of Alloys and Compounds, 453(1-2), 298–303. DOI:10.1016/j.jallcom.2006.11.058
Downloads
Published
Issue
Section
License
Copyright (c) 2026 Oles Honchar Dnipro National University

This work is licensed under a Creative Commons Attribution 4.0 International License.
- Authors reserve the right of attribution for the submitted manuscript, while transferring to the Journal the right to publish the article under the Creative Commons Attribution License. This license allows free distribution of the published work under the condition of proper attribution of the original authors and the initial publication source (i.e. the Journal)
- Authors have the right to enter into separate agreements for additional non-exclusive distribution of the work in the form it was published in the Journal (such as publishing the article on the institutional website or as a part of a monograph), provided the original publication in this Journal is properly referenced
- The Journal allows and encourages online publication of the manuscripts (such as on personal web pages), even when such a manuscript is still under editorial consideration, since it allows for a productive scientific discussion and better citation dynamics (see The Effect of Open Access).

