THE QUANTUM-CHEMICAL ASPECTS OF STRUCTURING IN THERMAL CURTIUS REARRANGEMENT MECHANISM FOR SOME ARYL- AS WELL AS HETARYLACYL AZIDES: INFLUENCE OF ACID-CATALYTIC AND SOLVATION EFFECTS OF MEDIUM
DOI:
https://doi.org/10.15421/jchemtech.v34i1.336743Keywords:
ab initio calculations, thermal Curtius rearrangement, activation barrier, «ortho-effect» of а substituent, acid catalysis, solvation effectAbstract
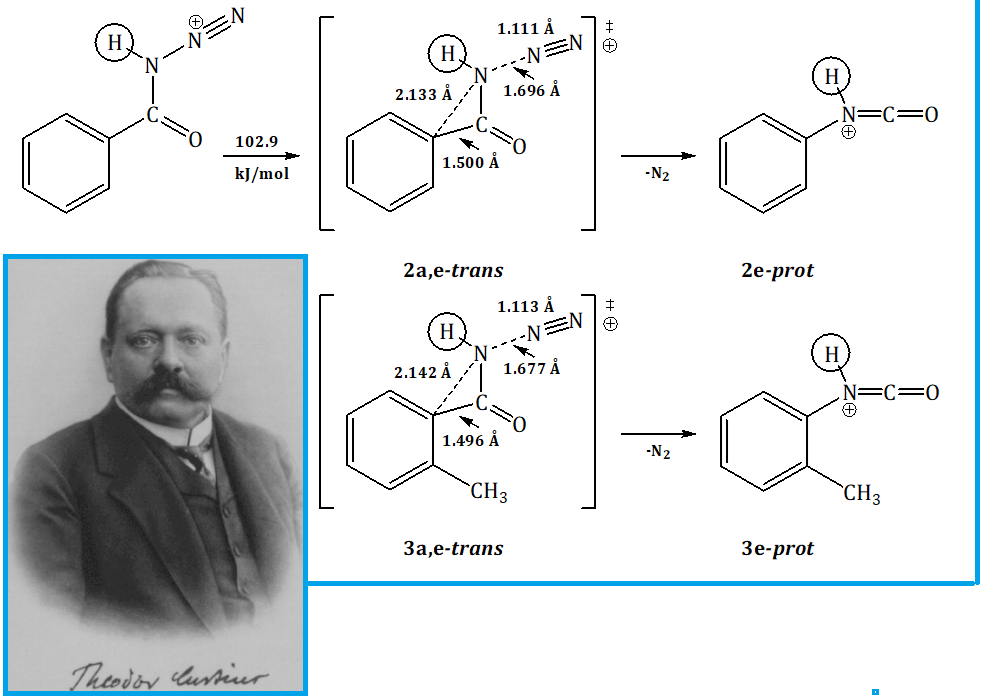
Using ab initio density functional method at the PBE1PBE/6-31G(d,p) level of theory, the features of the tautomeric equilibrium of open syn-forms for some aryl- as well as hetarylacyl azides have been investigated, which turned out to be more stable than the corresponding cyclic forms containing an oxatriazole fragment. Further analysis of individual sections for the thermal Curtius rearrangement potential energy surfaces using the model compound 2,6-dimethylpyridine-3,5-diacyl diazide as an example demonstrated the presence of a reaction pathway corresponding to the stepwise transformation of the syn,syn-form for the starting compound into the corresponding diisocyanate as the final reaction product. In this case, the sequential transformation of both acyl azide groups has been accompanied by intramolecular migration of the hetaryl fragment with simultaneous cleavage of the nitrogen molecule at each of the reaction stages. The results of calculations are in good agreement with data of this type obtained earlier and indicate a low sensitivity of the reaction to structural modifications of the substrate, among which the greatest contribution is made by the manifestations of the "ortho-effect" of methyl substituents in the aromatic fragment of the molecules. The overall increase in the reactivity of the studied systems is also shown under the acid catalysis conditions with simultaneous consideration of nonspecific solvation effects of the benzene solution. This approach allows for the detailed conditions of the target transformation and can be actively used to predict the behavior of related systems within the selected computational approximation.
References
Maurya, A. K., de Souza, F. M., Gupta, R. K. (2023). Polyurethane and Its Composites: Synthesis to Application. In: Gupta, R. K. (ed). Polyurethanes: Preparation, Properties, and Applications. Vol. 1: Fundamentals. ACS Symposium Series, 1452, 1–20. Washington, DC. doi: 10.1021/bk-2023-1452.ch001
Tokar, A., Chihvintseva, O., Milosavljević, M. (2023). The Quantum-Chemical Prediction for Some NMR Spectral Parameters in Systems Based on Aromatic Polyamides. In: Karabegovic, I., Kovačević, A., Mandzuka, S. (eds). New Technologies, Development and Application VI. NT 2023. Lecture Notes in Networks and Systems, 707, 236–241. Springer, Cham. doi: 10.1007/978-3-031-34721-4_26
Tokar, A., Chihvintseva, O., Mirjanić, D. (2024). The Quantum-Chemical Aspects of Structuring for Some Aramide-Type Polymer Systems with Hetaryl Fragments. In: Karabegovic, I., Kovačević, A., Mandzuka, S. (eds). New Technologies, Development and Application VII. NT 2024. Lecture Notes in Networks and Systems, 1070, 589–596. Springer, Cham. doi: 10.1007/978-3-031-66271-3_63
Ghosh, A. K., Sarkar, A., Brindisi, M. (2018). The Curtius rearrangement: Mechanistic insight and recent applications in natural product syntheses. Org. Biomol. Chem., 16, 2006–2027. doi: 10.1039/C8OB00138C
Wu, Z., Zeng, X. (2022). Curtius-Type Rearrangement of Sulfinyl Azides: A Matrix Isolation and Computational Study. J. Phys. Chem. A., 126(27), 4367–4375. doi: 10.1021/acs.jpca.2c02469
McCulla, R. D., Gohar, G. A., Hadad, C. M., Platz, M. S. (2007). Computational Study of the Curtius-like Rearrangements of Phosphoryl, Phosphinyl, and Phosphinoyl Azides and Their Corresponding Nitrenes. J. Org. Chem., 72(25), 9426–9438. doi: 10.1021/jo0711687
Peng, X.-L., Ding, W.-L., Li, Q.-S., Li, Z.-S. (2017). Theoretical Insights into Photo-Induced Curtius Rearrangement of Chlorodifluoroacetyl Azide. Org. Chem. Front., 4, 1153–1161. doi: 10.1039/C7QO00083A
Xie, B.-B., Cui, Ch.-X., Fang, W.-H., Cui, G. (2018). Photoinduced Curtius rearrangements of fluorocarbonyl azide, FC(O)N3: a QM/MM nonadiabatic dynamics simulation. Phys. Chem. Chem. Phys., 20, 19363–19372. doi: 10.1039/C8CP02651C
Ruipérez, F. (2019). Application of quantum chemical methods in polymer chemistry. Int. Rev. Phys. Chem., 38(3–4), 343–403. doi: 10.1080/0144235X.2019.1677062
Frisch, M. J., Trucks, G. W., Schlegel, H. B., Scuseria, G. E., Robb, M. A., Cheeseman, J. R., Scalmani, G., Barone, V., Mennucci, B., Petersson, G. A., Nakatsuji, H., Caricato, M., Li, X., Hratchian, H. P., Izmaylov, A. F., Bloino, J., Zheng, G., Sonnenberg, J. L., Hada, M., Ehara, M., Toyota, K., Fukuda, R., Hasegawa, J., Ishida, M., Nakajima, T., Honda, Y., Kitao, O., Nakai, H., Vreven, T., Montgomery, J. A. Jr., Peralta, J. E., Ogliaro, F., Bearpark, M., Heyd, J. J., Brothers, E., Kudin, K. N., Staroverov, V. N., Keith, T., Kobayashi, R., Normand, J., Raghavachari, K., Rendell, A., Burant, J. C., Iyengar, S. S., Tomasi, J., Cossi, M., Rega, N., Millam, J. M., Klene, M., Knox, J. E., Cross, J. B., Bakken, V., Adamo, C., Jaramillo, J., Gomperts, R., Stratmann, R. E., Yazyev, O., Austin, A. J., Cammi, R., Pomelli, C., Ochterski, J. W., Martin, R. L., Morokuma, K., Zakrzewski, V. G., Voth, G. A., Salvador, P., Dannenberg, J. J., Dapprich, S., Daniels, A. D., Farkas, O., Foresman, J. B., Ortiz, J. V., Cioslowski, J., Fox, D. J. (2013). Gaussian 09 (Revision D.01). Gaussian Inc., Wallingford CT.
Merrick, J. P., Moran, D., Radom, L. (2007). An Evaluation of Harmonic Vibrational Frequency Scale Factors. J. Phys. Chem. A., 111(45), 11683–11700. doi: 10.1021/jp073974n
Tomasi, J. (2011). Selected features of the polarizable continuum model for the representation of solvation. WIREs Comput. Mol. Sci., 1(5), 855–867. doi: 10.1002/wcms.54
Ge, Y., Le, A., Marquino, G. J., Nguyen, P. Q., Trujillo, K., Schimelfenig, M., Noble, A. (2019). Tools for Prescreening the Most Active Sites on Ir and Rh Clusters toward C-H Bond Cleavage of Ethane: NBO Charges and Wiberg Bond Indexes. ACS Omega, 4(20), 18809–18819. doi: 10.1021/acsomega.9b02813
Tokar, A. V., Chigvintseva, O. P. (2024). [Theoretical study of thermal Curtius rearrangement mechanism for some aryl- as well as hetarylacyl azides with prediction of the following cyclotrimerization progress]. J. Chem. Technol., 32(4), 895–902 (in Ukrainian). doi: 10.15421/jchemtech.v32i4.310213
Harper, L. K., Shoaf, A. L., Bayse, C. A. (2015). Predicting Trigger Bonds in Explosive Materials through Wiberg Bond Index Analysis. ChemPhysChem, 16(18), 3886–3892. doi: 10.1002/cphc.201500773
Godara, S., Radhakrishnan, A., Paranjothy, M. (2020). Chemical Dynamics Simulations of Curtius Reaction of Acetyl- and Fluorocarbonyl Azides. J. Phys. Chem. A., 124(32), 6438–6444. doi: 10.1021/acs.jpca.0c04366
Nouri, A., Zahedi, E., Ehsani, M., Nouri, A., Balali, E. (2018). Understanding the kinetics and molecular mechanism of the Curtius rearrangement of 3-oxocyclobutane-1-carbonyl azide. Comput. Theor. Chem., 1130, 121–129. doi: 10.1016/j.comptc.2018.03.019
Kakkar, R., Zaidi, S., Grover, R. (2009). The Curtius Rearrangement of Some Organic Azides: A DFT Mechanistic Study. Int. J. Quantum Chem., 109(5), 1058–1069. doi: 10.1002/qua.21911
Abu-Eittah, R. H., Hassan, W. M. I., Zordok, W. (2015). A theoretical study of the thermal Curtius rearrangement of some cinnamoyl azides using the DFT approach. J. Struct. Chem., 56(4), 628–641. doi: 10.1134/S0022476615040046
Tarwade, V., Dmitrenko, O., Bach, R. D., Fox, J. M. (2008). The Curtius Rearrangement of Cyclopropyl and Cyclopropenoyl Azides. A Combined Theoretical and Experimental Mechanistic Study. J. Org. Chem., 73(21), 8189–8197. doi: 10.1021/jo801104t
Taherian, R., Chahkandi, B., Zahedi, E. (2021). A comprehensive theoretical analysis of Curtius rearrangement of syn-syn and syn-anti conformers of oxalyl diazide. J. Mol. Graphics Modell., 109, 108012. doi: 10.1016/j.jmgm.2021.108012
Tokar, A. V. (2024). [Thesaurus in Polymer Chemistry]. Dnipro, Ukraine: LIRA (in Ukrainian).
Kakkar, R., Arora, R., Zaidi, S. (2017). DFT studies on the acid-catalyzed Curtius reaction: the Schmidt reaction. Struct. Chem., 28(6), 1743–1756. doi: 10.1007/s11224-017-0952-9
Wolf, M. E., Vandezande, J. E., Schaefer, H. F. (2021). Catalyzed Reaction of Isocyanates (RNCO) with Water. Phys. Chem. Chem. Phys., 23, 18535–18546. doi: 10.1039/D1CP03302F
Kishi, V., Chahkandi, B., Zahedi, E., Allameh, S. (2024). A theoretical assessment of Curtius rearrangement of malonyl azide: Molecular mechanism insight and solvent effects. J. Mol. Liq., 396, 124078. doi: 10.1016/j.molliq.2024.124078
Downloads
Published
Issue
Section
License
Copyright (c) 2026 Oles Honchar Dnipro National University

This work is licensed under a Creative Commons Attribution 4.0 International License.
- Authors reserve the right of attribution for the submitted manuscript, while transferring to the Journal the right to publish the article under the Creative Commons Attribution License. This license allows free distribution of the published work under the condition of proper attribution of the original authors and the initial publication source (i.e. the Journal)
- Authors have the right to enter into separate agreements for additional non-exclusive distribution of the work in the form it was published in the Journal (such as publishing the article on the institutional website or as a part of a monograph), provided the original publication in this Journal is properly referenced
- The Journal allows and encourages online publication of the manuscripts (such as on personal web pages), even when such a manuscript is still under editorial consideration, since it allows for a productive scientific discussion and better citation dynamics (see The Effect of Open Access).

