ACID-BASE AND ELECTROCHEMICAL BEHAVIOR OF “SULFUR DIOXIDE – 1,3,5-TRIS-(2-HYDROXYETHYL)HEXAHYDROTRIAZINE – WATER” SOLUTIONS
DOI:
https://doi.org/10.15421/jchemtech.v34i1.338209Keywords:
sulfur dioxide, monoethanolamine, formaldehyde, 1,3,5-tris-(2-hydroxyethyl)hexahydrotriazine, acid-base interaction, specific electrical conductivityAbstract
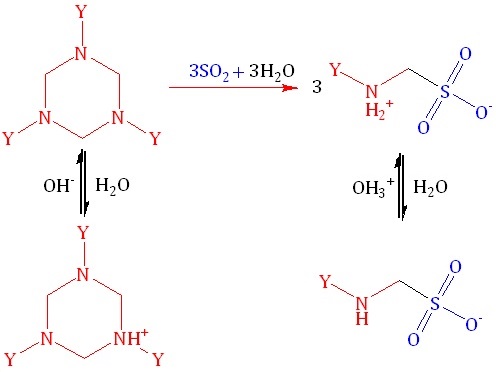
Establishing the features of acid-base and electrochemical behavior in “sulfur dioxide – 1,3,5-tris-(2-hydroxyethyl)hexahydrotriazine – water” solutions is undoubtedly an actual and important task. A pH-, redox-, and conductometric studies of protolytic equilibria were carried out for the solutions containing 1,3,5-tris-(2-hydroxyethyl)hexahydrotriazine (TZ; the reaction product of monoethanolamine with formaldehyde interaction), its protonated form (TZH+), N-(2-hydroxyethyl)aminomethanesulfonic acid (HEAMSA), and its anion, N-(2-hydroxyethyl)aminomethane-sulfonate (HEAMS) at a constant total content of amine nitrogen 0.1 mol/L with varying content of absorbed sulfur dioxide 0 £ QSO2 £ 0.12 mol/L at temperature range 273–313 K. It was shown that the addition of formaldehyde to solutions of “sulfur dioxide – monoethanolamine – water” leads to a decrease in their pH by 0.09–4.75 units, which is obviuosly caused by the formation of HEAMSA. The symbiotic nature of the change in DpH values with temperature (SO2 : N < 1.0 : 2.0) is noted at the same content of components in the solutions. Conductometry data indicate the association of free monoethanolamine, its ammonium sulfites and hydrosulfites into less mobile TZ, TZH+, HEАМSA and HEAMS–. Based on the developed mathematical model using pH-metry data, the ion-molecular component composition of “sulfur dioxide – 1,3,5-tris-(2-hydroxyethyl)hexahydrotriazine – water” solutions at 273–313 K was calculated. The concentration dependences on the ionic strength of the solutions were obtained. The concentration constants of TZ protonation and HEAMSA dissociation were estimated. It was shown that under the experimental conditions the thermodynamic dissociation constant of HEAMSA is practically independent on temperature and is equal to 10.14 ± 0.08. The obtained results are recommended to be used in the development of effective chemisorbents of sulfur dioxide.
References
Grygorenko, O. O., Biitseva, A. V., Zhersh, S. (2018). Amino sulfonic acids, peptidosulfonamides and other related compounds. Tetrahedron, 74(13), 1355-1421. https://doi.org/10.1016/j.tet.2018.01.033
Long, R. D., Hilliard, N. P., Chhatre, S. A. Timofeeva, T. V., Yakovenko, A. A., Dei, D. K., Mensah, E. A. (2010). Comparison of zwitterionic N-alkylaminomethanesulfonic acids to related compounds in the Good buffer series Beilstein J. Org. Chem. 6(31) https://doi.org/10.3762/bjoc.6.31
Hrydina, T.L., Khoma, R. E., Ennan, A. A.-A., Fedchuk, A.S., Hruzevskyi, O. A. (2019). [Investigations of the antimicrobial activity of aminomethanesulfonic acids against strains of Staphylococcus aureus with different antimicrobial susceptibility]. Zaporozhye Med. J., 21(2), 234-239. https://doi.org/10.14739/2310-1210.2019.2.161502 (in Ukrainian).
Khoma, R. E. (2019). [Acid-base interaction and sulfooxidation at chemosorption alkylamines aqueous solutions]. (Doctoral dissertation) http://ionc.com.ua/PDF/Khoma_thesis.pdf (in Ukrainian).
Khoma, R.E., Baumer, V.N., Antonenko, P.B., Snihach, A.O., Godovan, V.V., Ennan, A.A., Dlubovskii, R.M., Gelmboldt, V. V. (2019). Synthesis, crystal structure, and spectral characteristics of N-(n-propyl)aminomethanesulfonic acid. Acute toxicity of aminomethanesulfonic acid and its N-alkylated derivatives. Voprosy Khimii i Khimicheskoi Tekhnologii., (6), 255. https://doi.org/10.32434/0321-4095-2019-127-6-255-262
Khoma, R. E., Ennan, A. A.-A., Chebotaryov, A. N., Vodzinskii, S. V. (2019). [Aminomethansulfonic and alkylaminomethansulfonic buffer systems]. Ukr. Chem. J., 85(9), 3–16. https://doi.org/10.33609/0041-6045.85.9.2019.3-16 (in Russian)
Khoma, R. E., Ennan, A. A-A., Chebotaryov, A. N., Vodzinskii, S. V., Dlubovskii, R. M., Toporov, S. V. (2020). Electrochemical properties of aqueous solutions of sodium aminomethanesulfonates. Ukr. Chem. J., 86(11), 51–64. https://doi.org/10.33609/2708-129X.86.11.2020.51-64
Khoma, R., Ennan А. А.-А., Bienkovska, T. S., Osadchiy, L. T., Roy, E. L. (2021). Buffer systems based on aminometanesulphonate and monoethanolammo-nium N-alkylaminomethanes-ulphonates. Visn. Odes. nac. univ. Him., 26(2), 22–31. https://doi.org/10.18524/2304-0947.2021.2(78).233816
Zhao L., Zhou Y., Yao Q., Wang Y., Ge S., Liu X. (2021). Calcium Scale Inhibition of Stimulated Oilfield Produced Water Using Polyaspartic Acid/Aminomethanesulfonic Acid. ChemistrySelect. 6(15), 3692–3701. https://doi.org/10.1002/slct.202100853
Zhang M., Peng C., Zhu M., Yan W., Jiang H., Nan G., Li M., Zhou Z. (2024). Multifunctional Zwitterionic Modification of SnO2 in n–i–p Perovskite Solar Cells with Enhanced Fill Factor. ACS Sustainable Chem. Eng., 12(9). https://doi.org/10.1021/acssuschemeng.3c08004
Kondratenko, Y. A. (2024). From alkanolamines to protic alkanolammonium ionic liquids. J. Mol. Liq., 409, 125460. https://doi.org/10.1016/j.molliq.2024.125460
Deng T., Lv L., Li X., Wen J., Li H., Peng H., Chen H., Liu C., Bao L., Dang C., You Y., Chi F. (2025). Aminomethanesulfonic acid grafted polyamidoxime fibers with hydrophilicity, salt-tolerance and antimicrobial properties for highly efficient uranium extraction from seawater. Sep. Purif. Technol., 356A, 129610. https://doi.org/10.1016/j.seppur.2024.129610
Guo, A., Zhang, P., Ma, Y., Yuan, S. (2025). Molecular dynamics simulation of the electrochemical impact of aminomethanesulfonic acid on VO2+ in vanadium flow batteries. Mater. Today Chem. 47, 102815. https://doi.org/10.1016/j.mtchem.2025.102815
Zuwaid, H. A. B. (2025). High-Yield Cellulose Hydrolysis Using Silica-Amino Methanesulfonic Acid Catalyst: Hidrolisis Selulosa Hasil Tinggi Menggunakan Katalis Asam Metanesulfonat Silika-Amino. Academia Open. 10(1). https://doi.org/10.21070/acopen.10.2025.10869
Baker, M.V., Brown, D.H., Skelton, B.W., White, A.H. (1999). Chromium complexes of hydroxyl-functionalised 1,3,5-triazacyclohexanes. J. Chem. Soc., Dalton Trans. (9), 1483–1490. http://dx.doi.org/10.1039/a900122k
Akhmadali, K., Guzal, K., Ravshan, A., Muzaffar, A. (2019). Influence of the structure of aldehydes on the directions of their reactions with monoethanolamine. Chem. Chem. Eng., 2019(2), 5. https://doi.org/10.70189/1992-9498.1110
Fink, J. (2011). Petroleum Engineer's Guide to Oil Field Chemicals and Fluids. Saint Louis: Elsevier Science & Technology. https://doi.org/10.1016/C2009-0-61871-7
Kapoor, R., Selvaraju, S. B., Subramanian, V., Yadav, J. S. (2024). Microbial Community Establishment, Succession, and Temporal Dynamics in an Industrial Semi-Synthetic Metalworking Fluid Operation: A 50-Week Real-Time Tracking. Microorganisms, 12(2), 267. https://doi.org/10.3390/microorganisms12020267
Wylde, J., Taylor, G. (2019). Mercaptan scavenging revisited. Clariant Oil Services. https://www.clariant.com/-/media/Files/Business-Units/OMS/Oil-Services/Clariant-Article-in-Hydrocarbon-Engineering-2021-07-EN.pdf
Romero, I., Kucheryavskiy, S., Maschietti, M. (2021). Experimental study of the aqueous phase reaction of hydrogen sulfide with MEA-triazine using in situ Raman spectroscopy. Ind. Eng. Chem. Res., 60, 15549–15557. https://doi.org/10.1021/acs.iecr.1c03833
Romero I., Montero, F., Kucheryavskiy, S., Wimmer, R., Andreasen, A., Maschietti, M. (2023). Temperature- and pH-dependent kinetics of the aqueous phase hydrogen sulfide scavenging reactions with MEA-triazine. Ind. Eng. Chem. Res., 62, 8569-8580. https://doi.org/10.1021/acs.iecr.3c00668
Román M. N., Díaz M. A., Coll D. S. (2023). Study of the Reaction Mechanism of Triazines and Associated Species for H2S Scavenging. ACS Omega, 8(13), 12165–12172. https://doi.org/10.1021/acsomega.2c08103
Romero I., Montero F., Kucheryavskiy S., Wimmer R., Andreasen A., Maschietti M. (2023). Temperature- and pH-Dependent Kinetics of the Aqueous Phase Hydrogen Sulfide Scavenging Reactions with MEA-Triazine. Ind. Eng. Chem. Res., 62(21), 8269–8280. https://doi.org/10.1021/acs.iecr.3c00668
Tong, S., Zhu, J., Wang, Z., Yan, J. (2024). Highly Selective SO2 Capture by Triazine-Functionalized Triphenylamine-Based Nanoporous Organic Polymers. ACS Appl. Mater. Interfaces, 16(32), 42717–42725. https://doi.org/10.1021/acsami.4c08905
Pourebrahimi, S., Pirooz, M., Kazemeini, M., Vafajoo, L. (2024). Synthesis, characterization, and gas (SO2, CO2, NO2, CH4, CO, NO, and N2) adsorption properties of the CTF-1 covalent triazine framework-based porous polymer: experimental and DFT studies. J. Porous Mater. 31, 643–657. https://doi.org/10.1007/s10934-023-01538-9
Khoma, R. E. (2005) [Acid-base interaction of sulfur dioxide with amides aqueous solutions] (Unpublished habil. candidate's dissertation). A.V. Bogatsky Physico-Chemical Institute, Odesa, Ukraine (in Ukrainian).
Khoma, R. E., Tsyganenko, K. V., Bienkovska, T. S., Ishkov, Yu. V., Vodzinskii, S. V. (2025). Sulfur dioxide interaction with monoethanolammonium and polyethylenepolyammonium citrates aqueous solutions products composition and the relative stability. Ukr. Chem. J., 91(3), 3–24. https://doi.org/10.33609/2708-129X.91.3.2025.3-2
Wang, J., Huang, W., Xu, H., Cui, P., Qu, Z., Yan, N. (2023). High-efficient cyclic absorption of sulfur dioxide in Na-Mg-Ci3- compound system for wet flue gas desulfurization. Sep. Purif. Technol., 320, 124138. https://doi.org/10.1016/j.seppur.2023.124138
Vautherin, R., Métivier, H., Reguer, A., Benbelkacem, H. (2025). Use of a numerical model to evaluate SO2 absorption efficiency by sodium sulfite in packed and spray columns. Water Sci. Technol. 91(8), 907–922. https://doi.org/10.2166/wst.2025.049
Downloads
Published
Issue
Section
License
Copyright (c) 2025 Oles Honchar Dnipro National University

This work is licensed under a Creative Commons Attribution 4.0 International License.
- Authors reserve the right of attribution for the submitted manuscript, while transferring to the Journal the right to publish the article under the Creative Commons Attribution License. This license allows free distribution of the published work under the condition of proper attribution of the original authors and the initial publication source (i.e. the Journal)
- Authors have the right to enter into separate agreements for additional non-exclusive distribution of the work in the form it was published in the Journal (such as publishing the article on the institutional website or as a part of a monograph), provided the original publication in this Journal is properly referenced
- The Journal allows and encourages online publication of the manuscripts (such as on personal web pages), even when such a manuscript is still under editorial consideration, since it allows for a productive scientific discussion and better citation dynamics (see The Effect of Open Access).

