THERMODYNAMIC PROPERTIES OF 1-[1-(4-METHOXYPHENYL)-2-METHYL-5-PHENYL-PYRROLE-3-YL]ETHANONE SOLUTIONS IN ORGANIC SOLVENTS
DOI:
https://doi.org/10.15421/jchemtech.v34i1.338413Keywords:
solubility, enthalpy of dissolution, enthalpy of fusion, enthalpy of mixing, polysubstituted pyrrole derivatives, 1-[1-(4-methoxyphenyl)-2-methyl-5-phenyl-pyrrole-3-yl]ethanone, synthesis; IR spectraAbstract
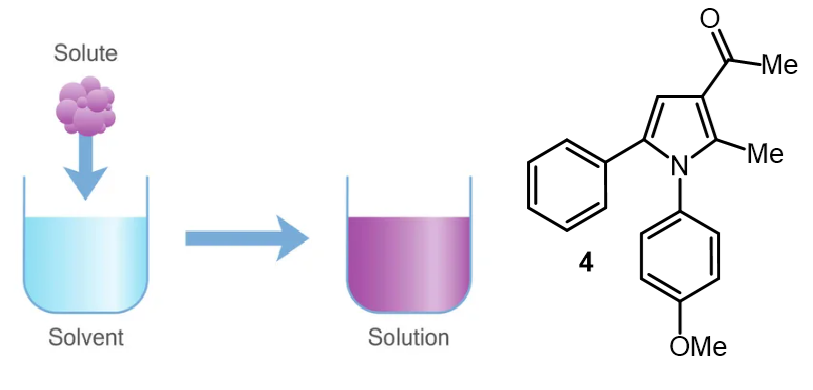
Using the Paal-Knorr reaction, 1-[1-(4-methoxyphenyl)-2-methyl-5-phenyl-pyrrole-3-yl]ethanone was produced. The main stages of synthesis and yield of the target compound are described. The temperature dependence of the solubility of the synthesized compound in the temperature range 275.70–299.40 K was investigated experimentally. The use of the gravimetric method for studying solubility at atmospheric pressure in solvents of different classes is described. Using the Van't Hoff equation, the obtained data were converted to standard molar enthalpies and entropies of dissolution. The enthalpy and entropy of fusion of the obtained substance were determined by the differential thermal method. The equations for converting the research results to standard conditions (298.15 K) are given, and the results are used to calculate the thermodynamic parameters of the mixing and dissolution process of the studied ketone compound with organic solvents. The solubility of 1-[1-(4-methoxyphenyl)-2-methyl-5-phenyl-pyrrole-3-yl]ethanone in all solvents increased with increasing temperature, and these data are in good agreement with the literature data for similar substances. The interaction characteristics of the synthesized substance with solvents of different polarity were established, and the dependencies of the thermal effects of dissolution were obtained. This study of the pyrrole derivative 1-[1-(4-methoxyphenyl)-2-methyl-5-phenyl-pyrrole-3-yl]ethanone is aimed at optimizing the synthesis processes of this compound, its further purification and processing for various uses.
References
Gomtsyan, A. (2012). Heterocycles in drugs and drug discovery. Chemistry of Heterocyclic Compounds, 48(1), 7–10. https://doi.org/10.1007/s10593-012-0960-z
Ganesh, B. H., Raj, A. G., Aruchamy, B., Nanjan, P., Drago, C., & Ramani, P. (2023). Pyrrole: A decisive scaffold for the development of therapeutic agents and structure‐activity relationship. ChemMedChem, 19(1), e202300447. https://doi.org/10.1002/cmdc.202300447
Vitaku, E., Smith, D. T., & Njardarson, J. T. (2014). Analysis of the structural diversity, substitution patterns, and frequency of nitrogen heterocycles among U.S. FDA approved pharmaceuticals. Journal of Medicinal Chemistry, 57(24), 10257–10274. https://doi.org/10.1021/jm501100b
Hunjan, M. K., Panday, S., Gupta, A., Bhaumik, J., Das, P., & Laha, J. K. (2021). Recent advances in functionalization of pyrroles and their translational potential. The Chemical Record, 21(4), 715–780. https://doi.org/10.1002/tcr.202100010
Burger, A. (1991). Isosterism and bioisosterism in drug design. Progress in Drug Research, 37, 288–362. https://doi.org/10.1007/978-3-0348-7139-6_7
Nakano, H., Umio, S., Kariyone, K., Tanaka, K., Kishimoto, T., & Noguchi, H. (1966). Total synthesis of pyrrolnitrin, a new antibiotic. Tetrahedron Letters, 7(7), 737–740. https://doi.org/10.1016/S0040-4039(00)90255-7
Brothers, P. J., & Senge, M. O. (2022). An introduction to porphyrins for the twenty‐first century. In Fundamentals of Porphyrin Chemistry. Wiley. https://doi.org/10.1002/9781119129301.ch1
Ivan, B.-C., Barbuceanu, S.-F., Hotnog, C. M., Anghel, A. I., Ancuceanu, R. V., Mihaila, M. A., ... Dumitrascu, F. (2022). New pyrrole derivatives as promising biological agents: Design, synthesis, characterization, in silico, and cytotoxicity evaluation. International Journal of Molecular Sciences, 23(16), Article 8854. https://doi.org/10.3390/ijms23168854
Li Petri, G., Spanò, V., Spatola, R., Holl, R., Raimondi, M.V., Barraja, P., & Montalbano, A. (2020). Bioactive pyrrole-based compounds with target selectivity. European Journal of Medicinal Chemistry, 208, 112783. https://doi.org/10.1016/j.ejmech.2020.112783
Long, L., Zhang, H., Zhou, Z., Duan, L., Fan, D., Wang, R., Zhu, W. (2024). Pyrrole-containing hybrids as potential anticancer agents: An insight into current developments and structure activity relationships. European Journal of Medicinal Chemistry, 273, 116470. https://doi.org/10.1016/j.ejmech.2024.116470
Ahmad, S., Alam, O., Naim, M. J., Shaquiquzzaman, M., Alam, M. M., & Iqbal, M. (2018). Pyrrole: An insight into recent pharmacological advances with structure activity relationship. European Journal of Medicinal Chemistry, 157, 527–561. https://doi.org/10.1016/j.ejmech.2018.08.002
Kostiuk, R. R., Horak, Y., & Sobechko, I. B. (2024). Thermodynamic parameters of a solution of 2 methyl 5 phenyl 1 (4 methylphenyl) pyrrole 3 carboxylic acid in alcohols. Chemistry, Technology and Application of Substances, 7(2), 1–6. https://doi.org/10.23939/ctas2024.02.001
Liu, Y., & Guo, H. (2021). Solubility determination and crystallization thermodynamics of an intermediate in different organic solvents. Journal of Molecular Liquids, 339, 116821. https://doi.org/10.1016/j.molliq.2021.116821
Paal, C. (1885). Synthese von Thiophen- und Pyrrolderivaten [Synthesis of thiophene and pyrrole derivatives]. Berichte der deutschen chemischen Gesellschaft, 18(1), 367–371. https://doi.org/10.1002/cber.18850180175
Knorr, L. (1885). [Action of diacetylsuccinic acid ester on ammonia and primary amine bases]. Berichte der deutschen chemischen Gesellschaft, 18(1), 299–311. https://doi.org/10.1002/cber.18850180154 (In German)
Dhananjaya, G., Rao, A. D., Hossain, K. A., Anna, V. R., & Pal, M. (2020). In silico studies and β-cyclodextrin mediated neutral synthesis of 4-oxo-4,5,6,7-tetrahydroindoles of potential biological interest. Tetrahedron Letters, 61(24), Article 151972. https://doi.org/10.1016/j.tetlet.2020.151972
Zhang, X., Weng, G., Zhang, Y., & Li, P. (2015). Unique chemoselective Paal–Knorr reaction catalyzed by MgI₂ etherate under solvent-free conditions. Tetrahedron, 71(18), 2595–2602. https://doi.org/10.1016/j.tet.2015.03.035
Kütt, A., Tshepelevitsh, S., Saame, J., Lõkov, M., Kaljurand, I., Selberg, S., & Leito, I. (2021). Strengths of acids in acetonitrile. European Journal of Organic Chemistry, 2021(9), 1407–1419. https://doi.org/10.1002/ejoc.202001649
Pliego Jr., J. R. (2024). Hybrid cluster–continuum method for single-ion solvation free energy in acetonitrile solvent. The Journal of Physical Chemistry A, 128(31), 6440–6449. https://doi.org/10.1021/acs.jpca.4c03593
Stenutz, R. (n.d.). Gutmann acceptor and donor number. https://www.stenutz.eu/chem/solv21.php
Shevchenko, D. S., Horak, Y. І., Matiichuk, V. V., Tischenko, N. I., Obushak, M. D., & Sobechko, I. B. (2025). Solubility study of 3-(1-(4-methylphenyl)-5-phenylpyrrol-2-yl)propanoic acid in organic solvents. Journal of Chemistry and Technologies, 33(1), 80–88. https://doi.org/10.15421/jchemtech.v33i1.312615
Maharana, A., & Sarkar, D. (2019). Solubility measurements and thermodynamic modeling of pyrazinamide in five different solvent-antisolvent mixtures. Fluid Phase Equilibria, 497, 33–54. https://doi.org/10.1016/j.fluid.2019.06.004
Sobechko, I., Chetverzhuk, Y., Horak, Y., Kochubei, V., & Velychkivska, N. (2017). Thermodynamic properties of 2-cyano-3-[5-(phenyl)-2-furyl]-2-propenamide and 2-cyano-3-[5-(4-methylphenyl)-2-furyl]-2-propenamide solutions in organic solvents. Chemistry & Chemical Technology, 11(2), 131–137. https://doi.org/10.23939/chcht11.02.131
Du, C. (2022). The solubility of ethyl candesartan in mono solvents and investigation of intermolecular interactions. Liquids, 2(4), 404–412. https://doi.org/10.3390/liquids2040023
Shevchenko, D. S., Horak, Y. І., Tischenko, N. I., Pyshna, D. B., Obushak, M. D., & Sobechko, I. B. (2025). Thermodynamic parameters of the solubility of 3-(1,5-diphenylpyrrol-2-yl)propanoic acid in organic solvents. Voprosy Khimii i Khimicheskoi Tekhnologii, 2(159), 24–32. https://doi.org/10.32434/0321-4095-2025-159-2-24-32
Klachko, O., Matiychuk, V., Sobechko, I., Serheyev, V., & Tishchenko, N. (2020). Thermodynamic properties of 6-methyl-2-oxo-4-aryl-1,2,3,4-tetrahydropyrimidine-5-carboxylic acid esters. Chemistry & Chemical Technology, 14(3), 277–283. https://doi.org/10.23939/chcht14.03.277
Filipa, A., & Ribeiro da Silva, M. A. V. (2014). Experimental and high level ab initio enthalpies of formation of di-, tri-, tetra- and pentamethyl-substituted pyrroles. The Journal of Chemical Thermodynamics, 75, 1–7. https://doi.org/10.1016/j.jct.2014.04.003
Acree, W., & Chickos, J. S. (2016). Phase transition enthalpy measurements of organic and organometallic compounds. Sublimation, vaporization and fusion enthalpies from 1880 to 2015. Part 1. C1–C10. Journal of Physical and Chemical Reference Data, 45(3), Article 033101. https://doi.org/10.1063/1.4948363
Sobechko, I., Horak, Y., Dibrivnyi, V., Obushak, M., & Goshko, L. (2019). Thermodynamic properties of 2-methyl-5-arylfuran-3-carboxylic acids chlorine derivatives in organic solvents. Chemistry & Chemical Technology, 13(3), 280–287. https://doi.org/10.23939/chcht13.03.280
Sobechko, I., Dibrivnyi, V., Horak, Y., Kochubei, V., & Obushak, M. (2017). Thermodynamic properties of solubility of 2-methyl-5-arylfuran-3-carboxylic acids in organic solvents. Chemistry & Chemical Technology, 11(4), 397–404. https://doi.org/10.23939/chcht11.04.397
Downloads
Published
Issue
Section
License
Copyright (c) 2025 Oles Honchar Dnipro National University

This work is licensed under a Creative Commons Attribution 4.0 International License.
- Authors reserve the right of attribution for the submitted manuscript, while transferring to the Journal the right to publish the article under the Creative Commons Attribution License. This license allows free distribution of the published work under the condition of proper attribution of the original authors and the initial publication source (i.e. the Journal)
- Authors have the right to enter into separate agreements for additional non-exclusive distribution of the work in the form it was published in the Journal (such as publishing the article on the institutional website or as a part of a monograph), provided the original publication in this Journal is properly referenced
- The Journal allows and encourages online publication of the manuscripts (such as on personal web pages), even when such a manuscript is still under editorial consideration, since it allows for a productive scientific discussion and better citation dynamics (see The Effect of Open Access).

