ANTIMICROBIAL COPOLYMERS FROM PARA-AMINOPYRIDINE METHACRYLATE AND METHYL METHACRYLATE: SYNTHESIS AND STRUCTURE–PROPERTY RELATIONSHIPS
DOI:
https://doi.org/10.15421/jchemtech.v34i1.341532Keywords:
p-aminopyridinemethacrylate, methyl methacrylate, copolymers, biocidal activity.Abstract
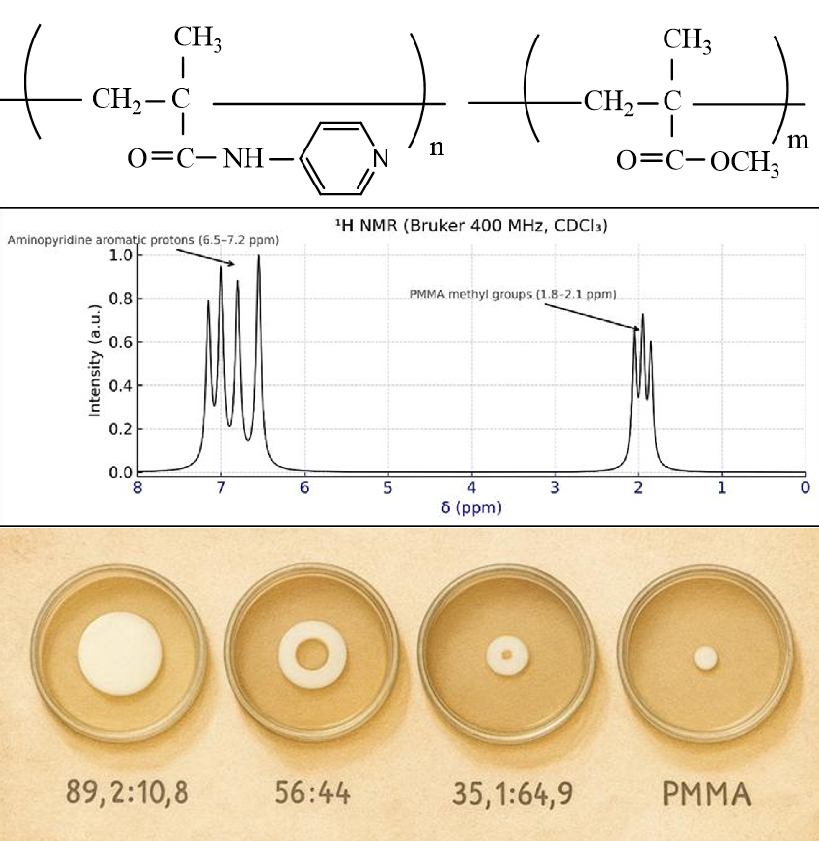
Functional copolymers based on para-aminopyridine methacrylate (p-APM) and methyl methacrylate (MMA) were synthesized via radical polymerization in benzene at 60 °C using azobisisobutyronitrile as the initiator. The copolymerization parameters determined by the Fineman–Ross method (r₁ = 0.85 ± 0.04, r₂ = 0.45 ± 0.03) indicated a higher reactivity of p-APM, resulting in copolymers enriched in aminopyridine units. IR and ¹H NMR analyses confirmed that polymerization proceeds through the vinyl groups with complete retention of amino and pyridine functionalities in the side chains. Thermogravimetric analysis revealed high thermal stability up to 380 °C, exceeding that of polymethyl methacrylate. Mechanical tests showed enhanced strength (91–95 MPa) and Vica softening temperatures of 148–152 °C, surpassing commercial Plexigum M-272. Antimicrobial activity, evaluated by the agar diffusion method, demonstrated pronounced inhibitory effects against both Gram-positive (Staphylococcus aureus) and Gram-negative (Escherichia coli) strains. Copolymers containing 89.2 mol% p-APM exhibited the highest antimicrobial efficiency, while moderate activity was observed at lower functional-monomer ratios. The biocidal efficiency correlated with both the content and distribution of p-APM units along the polymer chain. The combination of high thermal stability, mechanical strength, and antimicrobial activity suggests that p-APM–MMA copolymers are promising materials for biomedical coatings, membranes, and polymer systems with controlled antimicrobial properties.
References
Haq, I. U., Vieira, R. P., Lima, W. G., Lima, M. E., Krukiewicz, K. (2024). Antimicrobial polymers: elucidating the role of functional groups on antimicrobial activity. Arab Journal of Basic and Applied Science, 31, 325–344. https://doi.org/10.1080/25765299.2024.2366543
Alkarri, S., Saad, H. B., Soliman, M. (2024). On Antimicrobial Polymers: Development, Mechanism of Action, International Testing Procedures, and Applications. Polymers, 16(6), 771. https://doi.org/10.3390/polym16060771
Haktaniyan, M., Bradley, M. (2022). Polymers showing intrinsic antimicrobial activity. Chem. Soc. Rev., 51, 8584–8611. https://doi.org/10.1039 / D2CS00558A
Parcheta, M., Sobiesiak, M. (2023). Preparation and Functionalization of Polymers with Antibacterial Properties—Review of the Recent Developments. Materials (Basel), 16(12), 4411. https://doi.org/10.3390/ma16124411
Wu, S., Guo, W., Li, B., Zhou, H., Meng, H., Sun, J., Li, R., Guo, D., Zhang, X., Li, R., Qu, W. (2023). Progress of polymer-based strategies in fungal disease management: Designed for different roles. Front Cell Infect Microbial, 13, 1142029. https://doi.org/ 10.3389/fcimb.2023.1142029
Santoro, O., Izzo, L. (2024). Antimicrobial Polymer Surfaces Containing Quaternary Ammonium Centers (QACs): Synthesis and Mechanism of Action. Int. Mol. Sci., 25(14), 7587. https://doi.org/10.3390/ijms25147587
Salas-Ambrosio, P., Vexler, S., Sivasankaran, R., Vlahakis, N., Lai, R. S., Johnson, C., Baas-Maynard, S. I., Min, D. S., Lower, H., Doyle, A. G., Tang, Y., Rodriguez, J. A., Chen, I. A., Alaniz, J. R., Maynard, H. D. (2025). Biosourced Functional Hydroxybenzoate-co-Lactide Polymers with Antimicrobial Activity. Journal of the American Chemical Society, 147(22), 19230–19238. https://doi.org/10.1021/jacs.5c04624
Foster, L., Mizutani, M., Oda, Y., Palermo, E. F. (2017). Design and Synthesis of Amphiphilic Vinyl Copolymers with Antimicrobial Activity: Synthesis, Characterization, and Applications. In book: Polymers for Biomedicine, 243–272. https://doi.org/10.1002/9781118967904.ch8
Kumar, A., Jain, D., Srivastava, P., Nebhani, L. (2024). Solvent-Responsive Macroporous Polymer Gel Possessing Inherent Antimicrobial Activity Based on Quaternized 2-(Methyl(pyridine-4-yl)amino)ethyl Methacrylate. ACS Applied Polymer Materials, 6(17), 10218–10228. https://doi.org/10.1021/acsapm.4c01234
Yurtcan, S., Yolcu. Z. (2024). 4-Aminopyridine containing metal-2,6 -pyridine dicarboxylates and complex embedded hydrogels: Synthesis, characterization and antimicrobial applications. Inorganica Chimica Acta, 563, 121918. https://doi.org/10.1016/j.ica.2024.121918
Alamri, A., El-Newehy, M.H., Al-Deyab, S. S. (2012). Biocidal polymers: synthesis and antimicrobial properties of benzaldehyde derivatives immobilized onto amine-terminated polyacrylonitrile. Chemistry Central Journal, 6, 111. https://doi.org/10.1186/1752-153X-6-111
Dehar, M., Ould-Kada, S., Fortas Z., Dib-Bellahouel, S. (2016). Effect of the Chemical Structure of m and p N-Vinylbenzylidene of 5-Methyl-Thiazole and 1,2,4-Triazole on Antimicrobial Activity. Oriental Journal of Chemistry, 32(4), 2043–2049. http://dx.doi.org/10.13005/ojc/320431
Barman, S., Konai, M. M., Samaddar, S., Haldar J. (2019). Amino Acid Conjugated Polymers: Antibacterial Agents Effective against Drug-Resistant Acinetobacter baumannii with No Detectable Resistance. ACS Appl Mater Interfaces, 11(37), 33559–33572. http://doi.org/10.1021/acsami.9b09016
Tang, Z., Li, D., Liu, X., Wu, Z., Liu, W., Brash, J., Chen, H. (2013). Vinyl-monomer with lysine side chains for preparing copolymer surfaces with fibrinolytic activity. Polym. Chem., 4, 1583–1589. https://doi.org/10.1039/C2PY20944F
Nowak, M. G., Skwarecki, A. S., Milewska, M. J. (2021). Amino Acid Based Antimicrobial Agents – Synthesis and Properties. Chem Med Chem, 16(23), 3495–3625. https://doi.org/10.1002/cmdc.202100503
Marastoni, M., Trapella, C., Scotti, A., Fantinati, A., Ferretti, V., Marzola, E., Eleonora, G., Gavioli, R., Preti, D. (2017). Naphthoquinone amino acid derivatives, synthesis and biological activity as proteasome inhibitors. Journal of enzyme inhibition and medicinal chemistry, 32(1), 865–877. https://doi.org/10.1080/14756366.2017.1334649
Song, M.K., Lee, S. J., Kang, Y. Y., Lee, Y., Mok, H., Ahn, J. H. (2017). Biological synthesis and anti-inflammatory activity of arylalkylamine. Appl. Biol. Chem., 60(6), 597–602. https://doi.org/10.1007/s13765-017-0315-7
Deka, N., Bera, A., Roy, D., De, P. (2022). Methyl Methacrylate-Based Copolymers: Recent Developments in the Areas of Transparent and Stretchable Active Matrices. ACS Omega, 7(42), 36929–36944. https://doi.org/10.1021/acsomega.2c04564
Said, M. N. A., Hasbullah, N.A., Rosdi, M. R. H., Musa, M. S., Rusli, A., Ariffin, A., Shafiq, M. D. (2022). Polymerization and Applications of Poly(methyl methacrylate)–Graphene Oxide Nanocomposites: A Review. ACS Omega, 7(51), 47490–47503. https://doi.org/10.1021/acsomega.2c04483
Benadda, M., Oussadi, K., Haddou, B., Benettayeb, A., Lal, B., Ghosh, S. (2025). A green chemistry of the polymerization of methyl methacrylate (MMA) and a new copolymer of propylene oxide (PO) using natural catalysts. International Journal of polymer analysis and characterization, 30(4), 441–456. https://doi.org/10.1080/1023666X.2025.2474015
Abdulrazzaq, S. N., Jafarzadeh, T. S., Pourhajibagher, M., Behroozibakhsh, M., Masaeli, R., Bahador, A. (2018). Evaluation of Antimicrobial Properties of Conventional Poly(Methyl Methacrylate) Denture Base Resin Materials Containing Hydrothermally Synthesised Anatase TiO2 Nanotubes against Cariogenic Bacteria and Candida albicans. Iran J Pharm Res., 17(2), 161–172.
Juan Carlos, F., Rene, G. C. German, V.S., Susana, A.T. (2020). Antimicrobial Poly (methyl methacrylate) with Silver Nanoparticles for Dentistry: A Systematic Review. Appl. Sci., 10(11), 4007. https://doi.org/10.3390/app10114007
Giti, R., Zomorodian, K., Firouzmandi, M., Zareshahrabadi, Z., Rahmannasab, S. (2021). Antimicrobial Activity of Thermocycled Polymethyl Methacrylate Resin Reinforced with Titanium Dioxide and Copper Oxide Nanoparticles. İnternational Journal of Dentistry, 2021(1), 1–8. https://doi.org/10.1155/2021/6690806
Fineman, M., Ross, S.D. (1950). A Linear Method for Determining Monomer Reactivity Ratios in Copolymerization. Journal of Polymer Science, 5, 259–275.
Alfrey, T., Price, C.C. (1947). Relative reactivity of monomers in copolymerization. J. Polym. Sci., 2, 101–106.
Downloads
Published
Issue
Section
License
Copyright (c) 2026 Oles Honchar Dnipro National University

This work is licensed under a Creative Commons Attribution 4.0 International License.
- Authors reserve the right of attribution for the submitted manuscript, while transferring to the Journal the right to publish the article under the Creative Commons Attribution License. This license allows free distribution of the published work under the condition of proper attribution of the original authors and the initial publication source (i.e. the Journal)
- Authors have the right to enter into separate agreements for additional non-exclusive distribution of the work in the form it was published in the Journal (such as publishing the article on the institutional website or as a part of a monograph), provided the original publication in this Journal is properly referenced
- The Journal allows and encourages online publication of the manuscripts (such as on personal web pages), even when such a manuscript is still under editorial consideration, since it allows for a productive scientific discussion and better citation dynamics (see The Effect of Open Access).

