SYNTHESIS AND CHARACTERIZATION OF ZrO₂@Fe₃O₄@TSC MAGNETIC NANOCOMPOSITE FOR SUSTAINABLE REMEDIATION OF HEAVY METAL FROM CONTAMINATED WATER
DOI:
https://doi.org/10.15421/jchemtech.v34i1.342140Keywords:
Wastewater treatment, ZrO2@Fe3O4@TSC nanocomposite, TEM, VSM, XRD and FTIR.Abstract
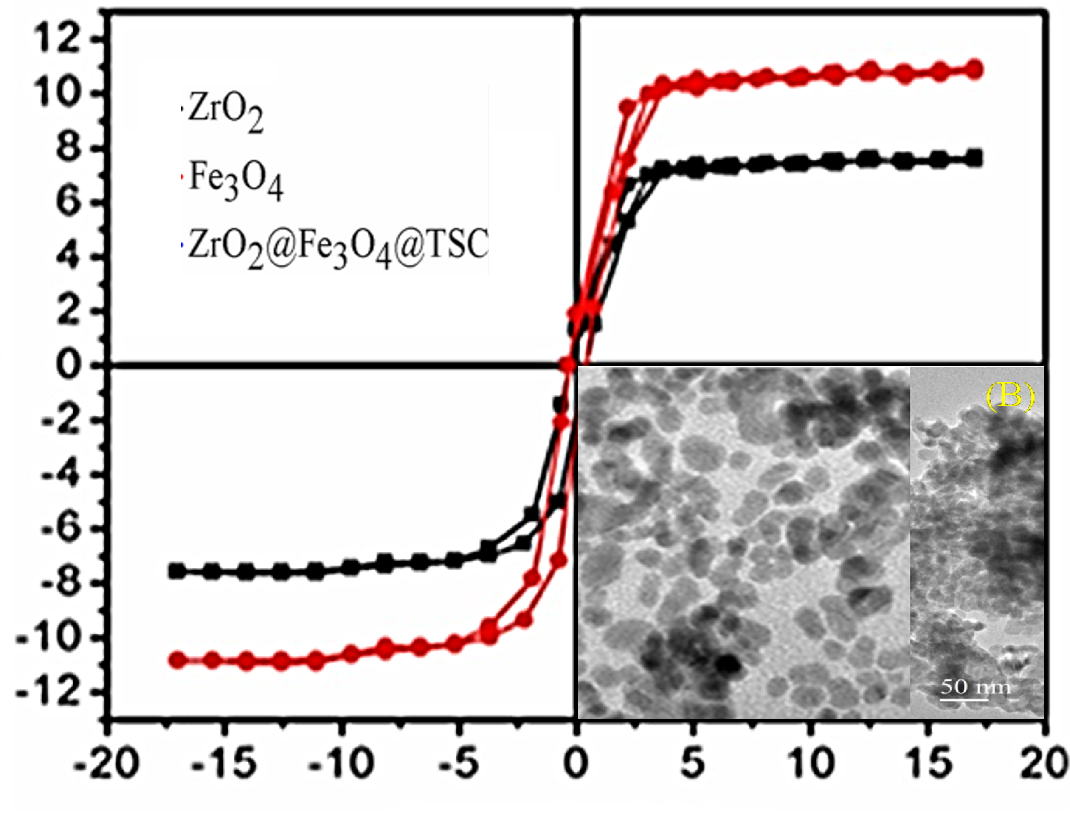
This study presents the synthesis and characterization of a novel adsorbent composed of iron oxide and zirconium oxide magnetic nanoparticles (ZrO2@Fe3O4 MNPs) functionalized with trisodium citrate (TSC). The structural and physicochemical properties of the ZrO2@Fe3O4@TSC nanocomposite were systematically investigated using Fourier-transform infrared spectroscopy (FTIR), X-ray diffraction (XRD), transmission electron microscopy (TEM) and vibrating sample magnetometry (VSM). The characterization results established that the ZrO2@Fe3O4@TSC nanocomposite shows a spherical morphology with particle sizes ranging from 16 to 20 nm. Magnetic measurements indicated that the nanocomposite possesses ferromagnetic behavior with a saturation magnetization value of 7.71 emu/g. The adsorption narration of the ZrO2@Fe3O4@TSC nanocomposite was evaluated for the removal of Cd (II) ions from aqueous solutions. The adsorption kinetics closely followed the pseudo-second-order model, demonstrating chemisorption as the leading process. The adsorption isotherm data were well described by the Langmuir isotherm model, suggesting monolayer adsorption on a homogenous surface. The maximum adsorption capacity was determined to be 50.26 mg/g at 303 K. These findings highlight the potential of ZrO2@Fe3O4@TSC nanocomposites as efficient adsorbents for heavy metal remediation in wastewater treatment applications.
References
Abdelfattah, I., & El-Shamy, A. M. (2024). A comparative study for optimizing photocatalytic activity of TiO2-based composites with ZrO2, ZnO, Ta2O5, SnO, Fe2O3, and CuO additives. Scientific Reports, 14(1), 27175. https://doi.org/10.1038/s41598-024-77752-5
Al-Mamun, M. R., Kader, S., Islam, M. S., & Khan, M. Z. H. (2019). Photocatalytic activity improvement and application of UV-TiO2 photocatalysis in textile wastewater treatment: A review. Journal of Environmental Chemical Engineering, 7(5), 103248. https://doi.org/10.1016/j.jece.2019.103248
Yang, J., Hou, B., Wang, J., Tian, B., Bi, J., Wang, N., Li, X., & Huang, X. (2019). Nanomaterials for the removal of heavy metals from wastewater. Nanomaterials, 9(3), 424. https://doi.org/10.3390/nano9030424
Putri, Y. E., Wendari, T. P., Dinda, D., Arnel, M., Faradilla, H., Refinel, R., & Efdi, M. (2024). The hydrothermal synthesis of SrTiO3 nanopolyhedral with the assistance of surfactants and their optical characteristics. Case Studies in Chemical and Environmental Engineering, 9, 100601. https://doi.org/10.1016/j.cscee.2023.100601
Mouden, A. E., Messaoudi, N. E., Guerraf, A. E., Bouich, A., Mehmeti, V., Lacherai, A., Jada, A., Americo-Pinheiro, J.H.P. (2023). Removal of cadmium and lead ions from aqueous solutions by noveldolomite-quartz@Fe3O4 nanocomposite fabricated as nanoadsorbent. Environmental Research, 225, 115606. https://doi.org/10.1016/j.envres.2023.115606
Haounati, R., Ouachtak, H., Haouti, R. E., Akhouairi, S., Largo, F., Akbal, F., Benlhachemi, A., Jada, A., Addi, A. A. (2020). Elaboration and properties of a new SDS/CTAB@Montmorillonite organoclay composite as a superb adsorbent for the removal of malachite green from aqueous solutions. Separation and Purification Technology, 255, 117335. https://doi.org/10.1016/j.seppur.2020.117335
Almeida, C. A. P., Debacher, N. A., Downs, A. J., Cottet, L., & Mello, C. A. D. (2009). Removal of methylene blue from colored effluents by adsorption on montmorillonite clay. Journal of Colloid and Interface Science, 332(1), 46–53. https://doi.org/10.1016/j.jcis.2008.12.012
de Resende, V. G., Garcia, F. L., Peigney, A., De Grave, E., & Laurent, C. (2009). Synthesis of Fe-ZrO2 nanocomposite powders by reduction in H2 of a nanocrystalline (Zr,Fe)O2 solid solution. Journal of Alloys and Compounds, 471(1-2), 204–210. https://doi.org/10.1016/j.jallcom.2008.03.045
Wahba, M. A., Mohamed, W. A. A., & Hanna, A. A. (2016). Sol-gel synthesis, characterization of Fe/ZrO2 nanocomposites and their photodegradation activity on indigo carmine and methylene blue textile dyes. International Journal of ChemTech Research, 9(6), 914–925.
Zeebaree, A. Y. S., Zeebaree, S. Y. S., Rashid, R. F., Zebari, O. I. H., Albarwry, A. J. S., Ali, A. F., Zebari, A. Y. S. (2022). Sustainable engineering of plant-synthesized TiO2 nanocatalysts: Diagnosis, properties and their photocatalytic performance in removing of methyleneblue dye from effluent. A review. Current Research in Green and Sustainable Chemistry, 5, 100312. https://doi.org/10.1016/j.crgsc.2022.100312
Daniel, S., Malathi, S., Balasubramanian, S., Sivakumar, M., & Sironmani, T. A. (2014). Multifunctional silver, copper and zero valent iron metallic nanoparticles for wastewater treatment. In Application of Nanotechnology in Water. John Wiley & Sons.
Cheng, Q., Yang, W., Chen, Q., Zhu, J., Li, D., Fu, L., Zhou, L. (2020). Fe-doped zirconia nanoparticles with highly negative conduction band potential for enhancing visible light photocatalytic performance. Applied Surface Science, 530, 147291. https://doi.org/10.1016/j.apsusc.2020.147291
Manyangadze, M., Chikuruwo, N. H. M., Narsaiah, T. B., Chakra, C. S., Radhakumari, M., & Danha, G. (2020). Enhancing adsorption capacity of nano-adsorbents via surface modification: A review. South African Journal of Chemical Engineering, 31, 25–32. https://doi.org/10.1016/j.sajce.2019.11.003
Vo, L. Q., Anh-Tuan, V., Le, T. D., Huynh, C. D., Tran, H. V. (2024). Fe3O4/Graphene oxide/chitosan nanocomposite: A smart nanosorbent for lead (II) ion removal from contaminated water. ACS Omega, 9(15), 17506–17517. https://doi.org/10.1021/acsomega.4c00486
Yasin, A. S., Mohamed, Y. A., Kim, D., Yoon, S., Ra, H., & Lee, K. (2021). Efficiency enhancement of electro-adsorption desalination using iron oxide nanoparticle-incorporated activated carbon nanocomposite. Micromachines, 12(10), 1148. https://doi.org/10.3390/mi12101148
Hui, Y., & Zhang, S. (2021). A facile synthesis of Fe-doped zirconium oxide nanoparticles for enhancement of Rhodamine B dye degradation. International Journal of Electrochemical Science, 16(6), 210653. https://doi.org/10.20964/2021.06.53
Sreelekha, N., Subramanyam, K., Reddy, D. A., Murali, G., Varma, K. R., & Vijayalakshmi, R. P. (2016). Efficient photocatalytic degradation of rhodamine-B by Fe doped CuS diluted magnetic semiconductor nanoparticles under the simulated sunlight irradiation. Solid State Sciences, 62, 71–81. https://doi.org/10.1016/j.solidstatesciences.2016.11.001
Zhao, H., Song, F., Su, F., Shen, Y., & Li, P. (2020). Removal of cadmium from contaminated groundwater using a novel silicon/aluminum nanomaterial: An experimental study. Archives of Environmental Contamination and Toxicology, 79, 324–337. https://doi.org/10.1007/s00244-020-00784-1
Liang, L., & Gong, P. (2020). Urban and air pollution: A multi-city study of long-term effects of urban landscape patterns on air quality trends. Scientific Reports, 10, 18618. https://doi.org/10.1038/s41598-020-74524-9
Sarwar, N., Bibi, F. U. N., Junaid, A., & Alvi, S. (2024). Impact of urbanization and human development on ecological footprints in OECD and non-OECD countries. Heliyon, 10(18), e38058. https://doi.org/10.1016/j.heliyon.2024.e38058
Siddiqua, A., Hahladakis, J. N., & Al-Attiya, W. A. K. A. (2022). An overview of the environmental pollution and health effects associated with waste landfilling and open dumping. Environmental Science and Pollution Research, 29, 58514–58536. https://doi.org/10.1007/s11356-022-21578-z
Manisalidis, I., Stavropoulou, E., Stavropoulos, A., & Bezirtzoglou, E. (2020). Environmental and health impacts of air pollution: A review. Frontiers in Public Health, 8, 14. https://doi.org/10.3389/fpubh.2020.00014
Münzel, T., Hahad, O., Daiber, A., & Landrigan, P. J. (2022). Soil and water pollution and human health: What should cardiologists worry about? Cardiovascular Research, 118(17), 3271–3273. https://doi.org/10.1093/cvr/cvac082
Mustafa, B. M., & Hassan, N. E. (2024). Water contamination and its effects on human health: A review. Journal of Geography, Environment and Earth Science International, 28(1), 38–49. https://doi.org/10.9734/JGEESI/2024/v28i1743
Khushbu, Gulati, R., Sushma, Kour, A., & Sharma, P. (2022). Ecological impact of heavy metals on aquatic environment with reference to fish and human health. Journal of Applied and Natural Science, 14(4), 1471–1484. https://doi.org/10.31018/jans.v14i4.3900
Perera, P. A. C. T., Kodithuwakku, S. P., Sundarabarathy, T. V., & Edirisinghe, U. (2015). Bioaccumulation of cadmium in freshwater fish: An environmental perspective. Insight Ecology, 4(1), 1–12. https://doi.org/10.5567/ECOLOGY-IK.2015.1.12
Naddafi, K., Nabizadeh, R., Saeedi, R., Mahvi, A. H., Vaezi, F., Yaghmaeian, K., Ghasri, A., & Nazmara, S. (2007). Biosorption of lead (II) and cadmium (II) by protonated Sargassum glaucescens biomass in a continuous packed bed column. Journal of Hazardous Materials, 147(3), 785–791. https://doi.org/10.1016/j.jhazmat.2007.01.082
Sun, J., You-Peng, D., Xu, J., Feng-Min, Z., Qi-Yuan, H., Min-Min, T., Liu, Y., Yang, J., Hong-Yan, L., Fu, L., & Zhao, H. (2024). [Title of the specific article needed]. Respiratory Research, 25, 19. https://doi.org/10.1186/s12931-024-02726-0
Ganguly, K., Levänen, B., Palmberg, L., Åkesson, A., & Lindén, A. (2018). Cadmium in tobacco smokers: A neglected link to lung disease? European Respiratory Review, 27(147), 170122. https://doi.org/10.1183/16000617.0122-2017
Barregard, L., Bergström, G., & Fagerberg, B. (2014). Cadmium, type 2 diabetes, and kidney damage in a cohort of middle-aged women. Environmental Research, 135, 311–316. https://doi.org/10.1016/j.envres.2014.09.017
Wu, M., Song, J., Zhu, C., Wang, Y., Yin, X., Huang, G., Zhao, K., Zhu, J., Duan, Z., & Su, L. (2017). Association between cadmium exposure and diabetes mellitus risk: A PRISMA-compliant systematic review and meta-analysis. Oncotarget, 8(68), 113129–113141. https://doi.org/10.18632/oncotarget.22820
Bernard, A. (2004). Renal dysfunction induced by cadmium: Biomarkers of critical effects. Biometals, 17(5), 519–523. https://doi.org/10.1023/B:BIOM.0000045731.75602.b9
Das, S., Sultana, K. W., Ndhlala, A. R., Mondal, M., & Chandra, I. (2023). Heavy metal pollution in the environment and its impact on health: Exploring green technology for remediation. Environmental Health Insights, 17, 11786302231201259. https://doi.org/10.1177/11786302231201259
Lv, Y., Wang, P., Huang, R., Liang, X., Wang, P., Tan, J., Chen, Z., Dun, Z., Wang, J., Jiang, Q., Wu, S., Ling, H., Li, Z., & Yang, X. (2017). Cadmium exposure and osteoporosis: A population-based study and benchmark dose estimation in Southern China. Journal of Bone and Mineral Research, 32(10), 1990–2000. https://doi.org/10.1002/jbmr.3151
Wang, M., Wang, X., Liu, J., Wang, Z., Jin, T., Zhu, G., & Chen, X. (2021). The association between cadmium exposure and osteoporosis: A longitudinal study and predictive model in a Chinese female population. Frontiers in Public Health, 9, 762475. https://doi.org/10.3389/fpubh.2021.762475
Genchi, G., Sinicropi, M. S., Lauria, G., Carocci, A., & Catalano, A. (2020). The effects of cadmium toxicity. International Journal of Environmental Research and Public Health, 17(11), 3782. https://doi.org/10.3390/ijerph17113782
Takeya, I., Etsuko, K., Yasushi, S., Mirei, U., Mitsuhiro, O., Hideaki, N., & Koji, N. (2005). Estimation of cumulative cadmium intake causing Itai-itai disease. Toxicology Letters, 159(2), 192–201. https://doi.org/10.1016/j.toxlet.2005.05.011
Zarei, R., Sabokbar, A., Zarif, B. R., Bayat, M., & Haghnazari, N. (2024). Efficient biosorption of Zn (II), Cd (II), and Pb (II) by Aspergillus brasiliensis in industrial wastewater coupled with electrochemical monitoring via sensor enhanced with modified silver nanoparticles. Environmental Science and Pollution Research. https://doi.org/10.1007/s11356-024-35471-4
Ahmad, I., Asad, R. U., Maryam, L., & Masood, M. (2024). Treatment methods for cadmium removal from wastewater. In Cadmium Toxicity in Water. Springer. https://doi.org/10.1007/978-3-031-54005-9_7
Kim, J. J., Lee, S. S., Fenter, P., Myneni, S. C. B., Nikitin, V., & Peters, C. A. (2023). Carbonate coprecipitation for Cd and Zn treatment and evaluation of heavy metal stability under acidic conditions. Environmental Science & Technology, 57(8), 3104–3113. https://doi.org/10.1021/acs.est.2c07678
Sunil, K., Karunakaran, G., Yadav, S., Padaki, M., Zadorozhnyy, V., & Pai, R. K. (2018). Al-Ti2O6 a mixed metal oxide based composite membrane: A unique membrane for removal of heavy metals. Chemical Engineering Journal, 348, 671–684. https://doi.org/10.1016/j.cej.2018.05.017
Khan, Z., Elahi, A., Bukhari, D. A., & Rehman, A. (2022). Cadmium sources, toxicity, resistance and removal by microorganisms – A potential strategy for cadmium eradication. Journal of Saudi Chemical Society, 26(6), 101569. https://doi.org/10.1016/j.jscs.2022.101569
Goswami, S., Aich, K., Das, S., Das, A. K., Manna, A., & Halder, S. (2013). A highly selective and sensitive probe for colorimetric and fluorogenic detection of Cd2+ in aqueous media. Analytical Methods, 5(11), 2658–2663. https://doi.org/10.1039/c3an36884j
Peng, L., Chen, S., Song, H., Zheng, M., Luo, S., & Tie, B. (2023). Quick removal of suspended cadmium from irrigation water using water hyacinth (Eichhornia crassipes)-phosphoric fertilizer. Water, Air, & Soil Pollution, 234, 356. https://doi.org/10.1007/s11270-023-06365-x
Vasudevan, S., & Lakshmi, J. (2011). Effects of alternating and direct current in electrocoagulation process on the removal of cadmium from water. Journal of Hazardous Materials, 192(1), 26–34. https://doi.org/10.1016/j.jhazmat.2011.04.081
Santiago-Martínez, M. G., Lira-Silva, E., Encalada, R., Pineda, E., Gallardo-Pérez, J. C., Zepeda-Rodriguez, A., ... & Jasso-Chávez, R. (2015). Cadmium accumulation and toxicity in the microalga Euglena gracilis: Role of glutathione and phytochelatins. Journal of Hazardous Materials, 288, 104–112. https://doi.org/10.1016/j.jhazmat.2015.02.027
Asad, R. U., Masood, M., Maryam, L., Waqas, H., Raza, E., & Ahmad, I. (2025). Application of microbial technology for treatment of heavy metals contaminated wastewater. In Smart Waste and Wastewater Management by Biotechnological Approaches. Springer. https://doi.org/10.1007/978-981-97-8673-2_17
Bhagat, R., Walia, S. S., Kartik, S., Singh, R., Singh, G., & Hossain, A. (2024). The integrated farming system is an environmentally friendly and cost-effective approach to the sustainability of agri-food systems in the modern era of the changing climate: A comprehensive review. Food and Energy Security, 13(3), e534. https://doi.org/10.1002/fes3.534
Kuppan, N., Padman, M., Mahadeva, M., Srinivasan, S., & Devarajan, R. S. (2024). A comprehensive review of sustainable bioremediation techniques: Eco-friendly solutions for waste and pollution management. Waste Management Bulletin, 2(3), 154–171. https://doi.org/10.1016/j.wmb.2024.07.005
Bhattacharyya, K., Bhattacharjee, N., & Ganguly, S. (2023). Evidences for the augmented Cd (II) biosorption by Cd (II) resistant strain Candida tropicalis XTA1874 from contaminated aqueous medium. Scientific Reports, 13, 12034. https://doi.org/10.1038/s41598-023-38485-z
Naseer, A. (2024). Role of nanocomposites and nano-adsorbents for heavy metals removal and dyes. An overview. Desalination and Water Treatment, 320, 100662. https://doi.org/10.1016/j.dwt.2024.100662
Satyam, S., & Patra, S. (2024). Innovations and challenges in adsorption-based wastewater remediation: A comprehensive review. Heliyon, 10(5), e26573. https://doi.org/10.1016/j.heliyon.2024.e26573
Akhtar, M. S., Ali, S., & Zaman, W. (2024). Innovative adsorbents for pollutant removal: Exploring the latest research and applications. Molecules, 29(18), 4317. https://doi.org/10.3390/molecules29184317
Shahryari, T., Mostafavi, A., Afzali, D., & Rahmati, M. (2019). Enhancing cadmium removal by low-cost nanocomposite adsorbents from aqueous solutions; a continuous system. Composites Part B: Engineering, 173, 106963. https://doi.org/10.1016/j.compositesb.2019.106963
Sangvanich, T., Morry, J., Fox, C., Ngamcherdtrakul, W., Goodyear, S., Castro, D., & Yantasee, W. (2014). Novel oral detoxification of mercury, cadmium, and lead with thiol-modified nanoporous silica. ACS Applied Materials & Interfaces, 6(8), 5483–5493. https://doi.org/10.1021/am5007707
Alijani, H., Beyki, M. H., Fazli, Y., & Shariatinia, Z. (2017). A nanohybrid of mixed ferrite-polyaniline derivative copolymer for efficient adsorption of lead ions: Design of experiment for optimal condition, kinetic and isotherm study. Desalination and Water Treatment, 66, 338–345. https://doi.org/10.5004/dwt.2017.20196
Panigrahy, S. K., Nandha, A., Chaturvedi, M., Mishra, P. K. (2024). Novel nanocomposites with advanced materials and their role in waste water treatment. Next Sustainability, 4, 100042. https://doi.org/10.1016/j.nxsust.2024.100042
Nadaf, S., Jena, G. K., Rarokar, N., Gurav, N., Ayyanar, M., Prasad, S., & Gurav, S. (2023). Biogenic and biomimetic functionalized magnetic nanosystem: Synthesis, properties, and biomedical applications. Hybrid Advances, 3, 100038. https://doi.org/10.1016/j.hybadv.2023.100038
Gerasimov, E. (2022). Synthesis of nanocomposites and catalysis applications. Nanomaterials, 12(5), 731. https://doi.org/10.3390/nano12050731
Bhaskar, S., Awin, E. W., Kumar, K. C. H., Lale, A., Bernard, S., & Kumar, R. (2020). Design of nanoscaled heterojunctions in precursor derived t-ZrO2/SiOC(N) nanocomposites: Transgressing the boundaries of catalytic activity from UV to visible light. Scientific Reports, 10, 740. https://doi.org/10.1038/s41598-019-57394-8
Safari, J., Gandomi-Ravandi, S., & Haghighi, Z. (2016). Supported polymer magnets with high catalytic performance in the green reduction of nitroaromatic compounds. RSC Advances, 6(38), 31514–31525. https://doi.org/10.1039/c5ra26613k
Boamah, P. O., Huang, Y., Hua, M., Zhang, Q., Liu, Y., Onumah, J., Wang, W., & Song, Y. (2015). Removal of cadmium from aqueous solution using low molecular weight chitosan derivative. Carbohydrate Polymers, 122, 255–264. https://doi.org/10.1016/j.carbpol.2015.01.004
Kowanga, K. D., Gatebe, E., Mauti, G. O., & Mauti, E. M. (2016). Kinetic, sorption isotherms, pseudo-first-order model and pseudo-second-order model studies of Cu(II) and Pb(II) using defatted Moringa oleifera seed powder. The Journal of Phytopharmacology, 5(2), 71–78.
Nakhlestani, N., Taghavi, L., Dehghani, M., & Panahi, H. A. (2024). Efficient removal of Pb (II) and Cd (II) ions from aqueous solutions using a 5-aminoisophthalic acid polymerized magnetic graphene oxide composite. Iranian Journal of Chemistry and Chemical Engineering, 43(7), 2721–2736.
Raven, K. P., Jain, A., & Loeppert, R. H. (1998). Arsenite and arsenate adsorption on ferrihydrite: Kinetics, equilibrium and adsorption envelopes. Environmental Science & Technology, 32(3), 344–349. https://doi.org/10.1021/es970421p
Dada, A. O., Adekola, F. A., Odebunmi, E.O., Ogunlaja, A.S., & Bello, O. S. (2021). Two-three parameters isotherm modeling, kinetics with statistical validity, desorption and thermodynamic studies of adsorption of Cu(II) ions onto zerovalent iron nanoparticles. Scientific Reports, 11, 16454. https://doi.org/10.1038/s41598-021-95090-8
Langmuir, I. (1918). The adsorption of gases on plane surfaces of glass, mica and platinum. Journal of the American Chemical Society, 40(9), 1361–1403. https://doi.org/10.1021/ja02242a004
Li, Y. H., Ding, J., Luan, Z., Di, Z., Zhu, Y., Xu, C., Wu, D., & Wei, B. (2003). Competitive adsorption of Pb2+, Cu2+ and Cd2+ ions from aqueous solutions by multiwalled carbon nanotubes. Carbon, 41(14), 2787–2792. https://doi.org/10.1016/S0008-6223(03)00392-0
Wang, Y., Ye, G., Chen, H., Hu, X., Niu, Z., & Ma, S. (2015). Functionalized metal-organic framework as a new platform for efficient and selective removal of cadmium (II) from aqueous solution. Journal of Materials Chemistry A, 3(29), 15292–15298. https://doi.org/10.1039/c5ta03201f
Bai, L., Hu, H. P., Fu, W., Wan, J., Cheng, X., Zhuge, L., Xiong, L., & Chen, Q. (2011). Synthesis of a novel silica-supported dithiocarbamate adsorbent and its properties for the removal of heavy metal ions. Journal of Hazardous Materials, 195, 261–275. https://doi.org/10.1016/j.jhazmat.2011.08.038
Dubey, A., Mishra, A., & Singhal, S. (2014). Application of dried plant biomass as novel low-cost adsorbent for removal of cadmium from aqueous solution. International Journal of Environmental Science and Technology, 11, 1043–1050. https://doi.org/10.1007/s13762-013-0278-0
Guo, X., Du, B., Wei, Q., Yang, J., Hu, L., Yan, L., & Xu, W. (2014). Synthesis of amino functionalized magnetic graphenes composite material and its application to remove Cr(VI), Pb(II), Hg(II), Cd(II) and Ni(II) from contaminated water. Journal of Hazardous Materials, 278, 211–220. https://doi.org/10.1016/j.jhazmat.2014.05.075
Badruddoza, A. Z. M., Shawon, Z. B. Z., Daniel, T. W. J., Hidajat, K., & Uddin, M. S. (2013). Fe3O4/cyclodextrin polymer nanocomposites for selective heavy metals removal from industrial wastewater. Carbohydrate Polymers, 91(1), 322–332. https://doi.org/10.1016/j.carbpol.2012.08.030
Hua, R., & Li, Z. (2014). Sulfhydryl functionalized hydrogel with magnetism: Synthesis, characterization and adsorption behavior study for heavy metal removal. Chemical Engineering Journal, 249, 189–200. https://doi.org/10.1016/j.cej.2014.03.097
Lemessa, G., Chebude, Y., Demesa, A. G., Fadeev, E., Koiranen, T., & Alemayehu, E. (2024). Development of suitable magnetite–diatomite nanocomposite for Cd (II) adsorptive removal from wastewater. Scientific African, 24, e02213. https://doi.org/10.1016/j.sciaf.2024.e02213
Downloads
Published
Issue
Section
License
Copyright (c) 2026 Oles Honchar Dnipro National University

This work is licensed under a Creative Commons Attribution 4.0 International License.
- Authors reserve the right of attribution for the submitted manuscript, while transferring to the Journal the right to publish the article under the Creative Commons Attribution License. This license allows free distribution of the published work under the condition of proper attribution of the original authors and the initial publication source (i.e. the Journal)
- Authors have the right to enter into separate agreements for additional non-exclusive distribution of the work in the form it was published in the Journal (such as publishing the article on the institutional website or as a part of a monograph), provided the original publication in this Journal is properly referenced
- The Journal allows and encourages online publication of the manuscripts (such as on personal web pages), even when such a manuscript is still under editorial consideration, since it allows for a productive scientific discussion and better citation dynamics (see The Effect of Open Access).

