SYNTHESIS AND STUDY OF AN ADHESIVE COMPOSITION BASED ON OXALIC ACID, FORMALDEHYDE AND PHTHALIMIDE USING IR, UV SPECTROSCOPY AND DFT CALCULATIONS
DOI:
https://doi.org/10.15421/jchemtech.v34i1.344276Keywords:
environmentally friendly adhesive, oxalic acid,, formaldehyde, phthalimide, adhesive formulation, molecular structureAbstract
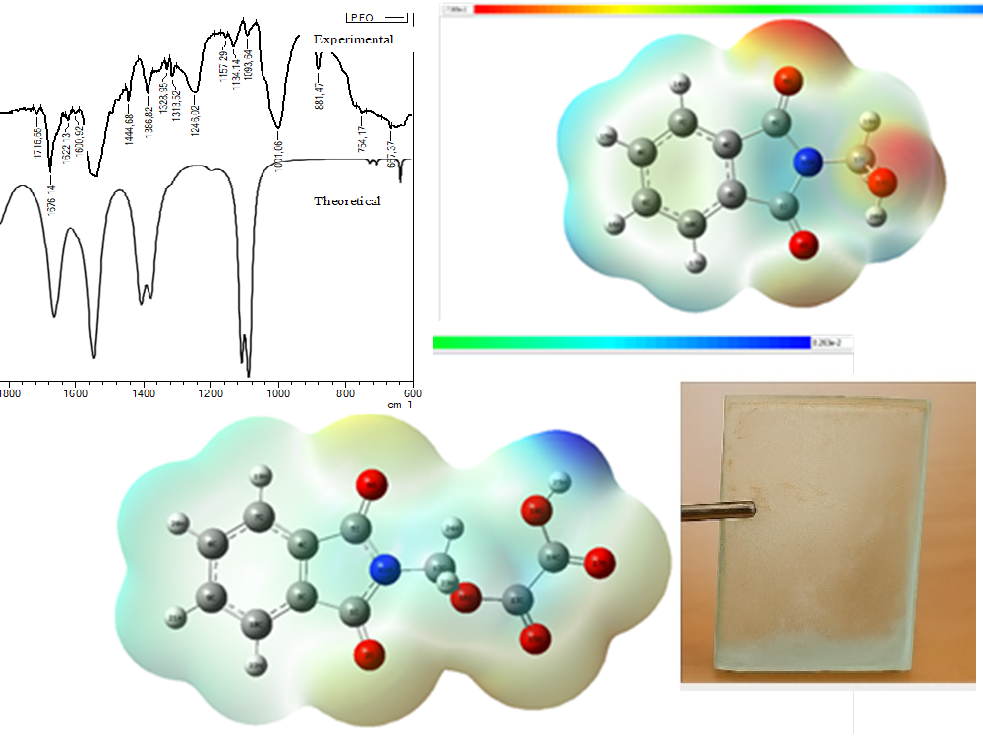
Developing environmentally friendly and highly effective adhesive compositions is a pressing challenge in modern materials science. This study synthesized and comprehensively characterized a new adhesive based on oxalic acid, formaldehyde, and phthalimide. These compounds were chosen as starting materials due to their expected adhesive properties and relative environmental friendliness. The resulting sample was analyzed using infrared (IR) and ultraviolet-visible (UV-Vis) spectroscopy to identify characteristic functional groups and intermolecular interactions. Additionally, a quantum-chemical analysis using density functional theory (DFT) was performed to evaluate the electronic structure, chemical bonding characteristics, and molecular stability. IR spectroscopy confirmed the presence of absorption bands corresponding to esterification and amide bonds, while UV-Vis spectroscopy data indicated the formation of conjugated systems that provide structural strength. Theoretical calculations are consistent with experimental observations and confirm the favorable electronic properties of the molecule. A combined experimental and theoretical approach provided a comprehensive understanding of the molecular architecture and potential adhesive properties. Thus, the adhesive based on oxalic acid, formaldehyde, and phthalimide exhibits good adhesion strength and is environmentally friendly to synthesize, making it promising for a wide range of industrial and sustainable applications.
References
Brockmann, W., Geiß, P. L., Klingen, J., & Schröder, K. B. (2009). Adhesive bonding: materials, applications and technology. John Wiley & Sons.
Marques, A. C., Mocanu, A., Tomić, N. Z., Balos, S., Stammen, E., Lundevall, A., Abrahami, S. T., Günther, R., de Kok, J. M. M., & Teixeira de Freitas, S. (2020). Review on adhesives and surface treatments for structural applications: Recent developments on sustainability and implementation for metal and composite substrates. Materials, 13(24), 5590. https://doi.org/10.3390/ma13245590.
Imam, S. H., Bilbao-Sainz, C., Chiou, B. S., Glenn, G. M., & Orts, W. J. (2013). Biobased adhesives, gums, emulsions, and binders: current trends and future prospects. Journal of Adhesion Science and Technology, 27(18-19), 1972–1997. https://doi.org/10.1080/01694243.2012.696892.
Maulana, S., Wibowo, E. S., Mardawati, E., Iswanto, A. H., Papadopoulos, A., & Lubis, M. A. R. (2024). Eco-friendly and high-performance bio-polyurethane adhesives from vegetable oils: a review. Polymers, 16(11), 1613. https://doi.org/10.3390/polym16111613.
Sancaktar E. (2018). Classification of adhesive and sealant materials. In Handbook of adhesion technology. Springer, Cham. https://doi.org/10.1007/978-3-319-55411-2_12.
Stoilova, A., Yordanov, D., Weiter, M., Georgiev, A., Zhivkov, I., & Dimov, D. (2019). Synthesis and photochromic properties of some N-phthalimide azo-azomethine dyes. A DFT quantum mechanical calculations on imine-enamine tautomerism and trans-cis photoisomerization. Spectrochimica acta. Part A, Molecular and biomolecular spectroscopy, 210, 230–244. https://doi.org/10.1016/j.saa.2018.11.033
Tenorio-Alfonso, A., Sánchez, M. C., & Franco, J. M. (2020). A review of the sustainable approaches in the production of bio-based polyurethanes and their applications in the adhesive field. Journal of Polymers and the Environment, 28, 749-774. https://doi.org/10.1007/s10924-020-01659-1.
Maulana, M. I., Lubis, M. A. R., Febrianto, F., Hua, L. S., Iswanto, A. H., Antov, P., Kristak, L., Mardawati, E., Sari, R. K., Zaini, L. H., Hidayat, W., Giudice, V. L., & Todaro, L. (2022). Environmentally friendly starch-based adhesives for bonding high-performance wood composites: a review. Forests, 13(10), 1614. https://doi.org/10.3390/f13101614.
Zeng, N., Xie, J. J., & Ding, C. (2012). Properties of the soy protein isolate/PVAc latex blend adhesives. Advanced Materials Research, 550, 1103–1107. https://doi.org/10.4028/www.scientific.net/AMR.550-553.1103.
Oliveira, J., Medeiros, E., De Barros, S., Azerêdo, M., Nunes, M., Tonoli, G., & Figueiredo, L. (2021). Environmentally friendly adhesives derived from glycerol-based polymers. Journal of Adhesion Science and Technology, 36, 98–108. https://doi.org/10.1080/01694243.2021.1915619.
Gadhave, R., Gadekar, P., & Mahanwar, P. (2017). Bio-Renewable Sources for Synthesis of Eco-Friendly Polyurethane Adhesives-Review, Open Journal of Polymer Chemistry, 7, 57–75. https://doi.org/10.4236/OJPCHEM.2017.74005.
Herrera, C., Ysinga, K. J., & Jenkins, C. L. (2019). Polysulfides synthesized from renewable garlic components and repurposed sulfur form environmentally friendly adhesives. ACS applied materials & interfaces, 11(38), 35312-35318. https://doi.org/10.1021/acsami.9b11204.
Wilker, J., & Román, J. (2019). Cooking Chemistry Transforms Proteins into High-Strength Adhesives. Journal of the American Chemical Society, 141(3), 1359–1365. https://doi.org/10.1021/jacs.8b12150.
Bartlett, M. D., & Crosby, A. J. (2014). High capacity, easy release adhesives from renewable materials. Advanced Materials (Deerfield Beach, Fla.), 26(21), 3405–3409. https://doi.org/10.1002/adma.201305593.
Imam, S. H., Gordon, S. H., Mao, L., & Chen, L. (2001). Environmentally friendly wood adhesive from a renewable plant polymer: characteristics and optimization. Polymer Degradation and Stability, 73(3), 529–533. https://doi.org/10.1016/S0141-3910(01)00114-8.
Wang, L., & Zhao, J. (2009). Encapsulation of chlorpyrifos in micro capsules prepared using industrial urea formaldehyde adhesive. Journal of Beijing University of Chemical Technology: Natural Science, 36(5), 46–51. https://journal.buct.edu.cn/EN/Y2009/V36/I5/46
Ugovšek, A., & Šernek, M. (2012). Curing of commercial phenol-formaldehyde adhesives. 64(11/12), 321–326. https://www.cabidigitallibrary.org/doi/full/10.5555/20133360847
Niyozov, E., Razzakov, K., Nazarov, S., Olimov, B., & Gafurova, G. (2024). Investigation of physicochemical properties of guanidine-based corrosion inhibitor. E3S Web of Conferences, 587, 03004. https://doi.org/10.1051/e3sconf/202458703004.
Schmidt, R., Frazier, C., & Laborie, M. (2000). Lab-Scale Synthesis of Isotopically Labeled Formaldehyde for the Production of Formaldehyde-Based Wood Adhesives, Holzforschung 54, 100–98. https://doi.org/10.1515/HF.2000.015.
Ram, S., Mehara, P., Kumar, A., Sharma, A. K., Chauhan, A. S., Kumar, A., & Das, P. (2022). Supported-Pd catalyzed carbonylative synthesis of phthalimides and isoindolinones using Oxalic acid as in situ CO surrogate with 2-iodobenzamides and 2-iodobenzylanilines in ppm-level catalyst loading. Molecular Catalysis, 530, 112606. https://doi.org/10.1016/j.mcat.2022.112606.
Amrullaev, A., Boltaeva, S., Rashitova, S., & Ganiev, B. (2024). Synthesis and study sorption properties oligo (poly)-mer sorbents based on urea-formaldehyde and cyanuric acid. In BIO Web of Conferences. EDP Sciences., 130, 06004. https://doi.org/10.1051/bioconf/202413006004.
Deng, S., Pizzi, A., Du, G., Zhang, J., & Zhang, J. (2014). Synthesis, structure, and characterization of glyoxal‐urea‐formaldehyde cocondensed resins. Journal of Applied Polymer Science, 131(21). https://doi.org/10.1002/app.41009.
Nemeş, N. S., & Negrea, A. (2023). Infrared and Visible Spectroscopy: Fourier Transform Infrared Spectroscopy and Ultraviolet–Visible Spectroscopy. Microbial Electrochemical Technologies: Fundamentals and Applications, 1, 163–200. https://doi.org/10.1002/9783527839001.ch6.
Nazarov, S., Amrieva, S., Ganiev, B., & Nazarov, N. (2024). Synthesis and spectroscopic study of adhesive polymer materials based on urea-formaldehyde and isoamyl alcohol. BIO Web of Conferences. EDP Sciences, 130, 06003. https://doi.org/10.1051/bioconf/202413006003.
Filatov, M., Lee, S., Nakata, H., & Choi, C. H. (2020). Computation of Molecular Electron Affinities Using an Ensemble Density Functional Theory Method. The journal of physical chemistry. A, 124(38), 7795–7804. https://doi.org/10.1021/acs.jpca.0c06976
Frisch, Michael J., Alice B. Nielsen. (2003). Gaussian 03 Programmer's Reference. Gaussian.
Hiscocks J., Frisch M. J. (2009). Gaussian 09: IOps Reference. – Wallingford, CT, USA: Gaussian.
Allouche, A. R. (2011). Gabedit—A graphical user interface for computational chemistry softwares. Journal of computational chemistry, 32(1), 174-182. https://doi.org/10.1002/jcc.21600.
Allouche, A. R. (2012). Gabedit is a free Graphical User Interface for computational chemistry packages. http://gabedit.sourceforge.net
Ganiev, B. Sh, Nazarov S.I, & Amrieva S.K. (2025). IR spectroscopy of glue based on oxalic acid, formaldehyde, and phthalimide. Abstract of the Uzbek-Tajik Symposium with International participation “Current state and prospects of polymer science development: synthesis, structure, properties and application”, 118–1120. https://doi.org/10.5281/zenodo.15194878
Krishnakumar, V., Balachandran, V., & Chithambarathanu, T. (2005). Density functional theory study of the FT-IR spectra of phthalimide and N-bromophthalimide. Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy, 62(4-5), 918–925. https://doi.org/10.1016/j.saa.2005.02.051.
Él’kin, M. D., Shal’nova, T. A., & Smirnov, A. P. (2010). Spectral manifestation of intermolecular interaction in phthalimide and isatin dimers. Journal of Applied Spectroscopy, 77, 22–27. https://doi.org/10.1007/s10812-010-9288-6.
Marciniak, A., Kotynia, A., Szkatuła, D., & Krzyżak, E. (2022). The 2-hydroxy-3-(4-aryl-1-piperazinyl)propyl Phthalimide Derivatives as Prodrugs—Spectroscopic and Theoretical Binding Studies with Plasma Proteins. International Journal of Molecular Sciences, 23(13), 7003. https://doi.org/10.3390/ijms23137003.
Husenov, K. S., Umarov, B. B., Turgunov, K. K., Ganiev, B. S., Mardonov, U. M., Ibragimov, B. T., Ashurov, J. M., & Safin, D. A. (2024). Synthesis, characterization, and computational studies of the zinc bromide complex with 2-amino-1,3,4-thiadiazole. Russian Journal of Inorganic Chemistry, 69(8), 1173–1183. https://doi.org/10.1134/S0036023624600941.
Dang, H., Wang, G., Yu, C., Ning, X., Zhang, J., Zhang, N., ... & Wang, C. (2021). Study on chemical bond dissociation and the removal of oxygen-containing functional groups of low-rank coal during hydrothermal carbonization: DFT calculations. ACS omega, 6(39), 25772–25781. https://doi.org/10.1021/acsomega.1c03866.
Khan, B. A., Hamdani, S. S., Khalid, M., Ashfaq, M., Munawar, K. S., Tahir, M. N., Braga, A. A. C., Shawky, A.M., Alqahtani, A. M., Abourehab, M. A. S., Gabr, G. A., Ibrahim, M. A. A., & Sidhom, P. A. (2023). Exploring Probenecid Derived 1,3,4-Oxadiazole-Phthalimide Hybrid as α-Amylase Inhibitor: Synthesis, Structural Investigation, and Molecular Modeling. Pharmaceuticals, 16(3), 424. https://doi.org/10.3390/ph16030424.
Benito, M., Barceló-Oliver, M., Frontera, A., & Molins, E. (2022). Oxalic Acid, a Versatile Coformer for Multicomponent Forms with 9-Ethyladenine. Crystals, 12(1), 89. https://doi.org/10.3390/cryst12010089
Downloads
Published
Issue
Section
License
Copyright (c) 2026 Oles Honchar Dnipro National University

This work is licensed under a Creative Commons Attribution 4.0 International License.
- Authors reserve the right of attribution for the submitted manuscript, while transferring to the Journal the right to publish the article under the Creative Commons Attribution License. This license allows free distribution of the published work under the condition of proper attribution of the original authors and the initial publication source (i.e. the Journal)
- Authors have the right to enter into separate agreements for additional non-exclusive distribution of the work in the form it was published in the Journal (such as publishing the article on the institutional website or as a part of a monograph), provided the original publication in this Journal is properly referenced
- The Journal allows and encourages online publication of the manuscripts (such as on personal web pages), even when such a manuscript is still under editorial consideration, since it allows for a productive scientific discussion and better citation dynamics (see The Effect of Open Access).

