BENZO[f][1,2,4]TRIAZINO[2,3-d][1,4]DIAZEPINES – A NEW HETEROCYCLIC SYSTEM: SYNTHESIS AND SPECTRAL CHARACTERISTICS
DOI:
https://doi.org/10.15421/jchemtech.v34i1.347428Keywords:
benzo[f][1,2,4]triazino[2,3-d][1,4]diazepines, acylation, cycle expansion, spectral data, X-Ray studyAbstract
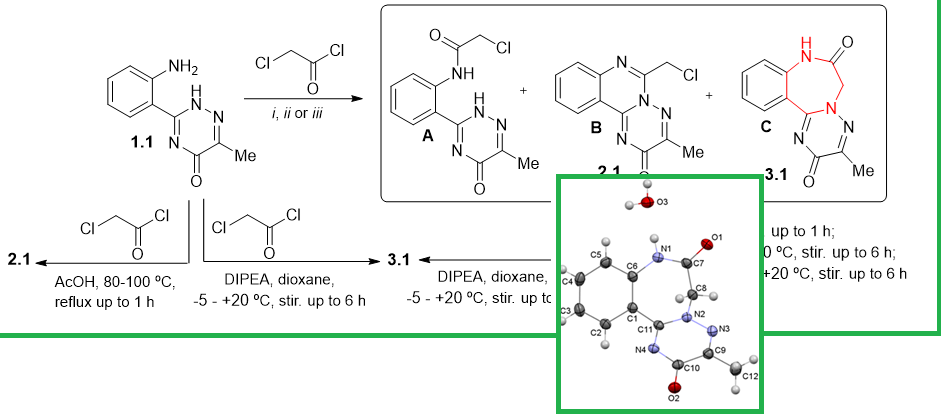
Benzodiazepines are an important group of heterocyclic compounds in organic and medicinal chemistry due to their wide range of biological activity and application in medical practice, significant potential for functionalization and synthesis of various compounds with fused cycles. In this work, we present simple methods for the synthesis of a new system of benzo[f]1,2,4]triazino[2,3-d][1,4]diazepines by acylation of of 3-(2-aminophenyl)-6-R-1,2,4-triazine-5(2H)-ones with chloroacetyl chloride. The peculiarities of the reaction, the optimal conditions for the synthesis of the new heterocyclic system, namely the ratio of reagents, solvents, temperature and duration of the reaction, have been established. It has been shown that the starting compounds containing donor-acceptor groups in the reaction in question form a mixture of two structural isomers. An alternative synthetic method has been developed, namely nucleophilic ring expansion of the corresponding 3-R-6-chloromethyl-2H-[1,2,4]triazino[2,3-c]quinazolin-2-ones, and the probable mechanism of this reaction has been discussed. Structural confirmation was achieved using ¹H and ¹³C NMR, LS-MS and X-ray.
References
Malki, Y., Martinez, J., & Masurier, N. (2021). 1,3-Diazepine: A privileged scaffold in medicinal chemistry. Medicinal Research Reviews, 41(4), 2247–2315. https://doi.org/10.1002/med.21795
Muhammad, A. R., Ashraf, A., Rehman, S. S., Shahid, S. A., Mahmood, A., & Faruq, M. (2019). 1,4-Diazepines: A review on synthesis, reactions and biological significance. Current Organic Synthesis, 16(5), 709–729. https://doi.org/10.2174/1570179416666190703113807
Salih, N., Salimon, J., & Hussien, H. (2021). Synthesis, characterization and in vitro antibacterial activity of novel 1,2,4-triazine and 1,2-diazepine derivatives. Biointerface Research in Applied Chemistry, 12(3), 3055–3065. https://doi.org/10.33263/BRIAC123.30553065
Maurya, H. K., Gautam, S. K., Pratap, R., Tandon, V. K., Kumar, A., Kumar, B., Saxena, S., Tripathi, D., Rajwanshi, M., Das, M., & Ram, V. J. (2014). Regioselective synthesis of polycyclic aza-oxa and aza-oxa-thia heteroarenes as Colo-205 and HepG2 carcinoma cells growth inhibitors. European Journal of Medicinal Chemistry, 81, 367–377. https://doi.org/10.1016/j.ejmech.2014.05.013
Tolu-Bolaji, O. O., Sojinu, S. O., Okedere, A. P., & Ajani, O. O. (2022). A review on the chemistry and pharmacological properties of benzodiazepine motifs in drug design. Arab Journal of Basic and Applied Sciences, 29(1), 287–306. https://doi.org/10.1080/25765299.2022.2117677
Zappala, M., Grasso, S., Micale, N., Polimeni, S., & De Micheli, C. (2001). Synthesis and structure–activity relationships of 2,3-benzodiazepines as AMPA receptor antagonists. Mini Reviews in Medicinal Chemistry, 1(3), 243–253. https://doi.org/10.2174/1389557013406783
Solyom, S., & Tarnawa, I. (2002). Non-competitive AMPA antagonists of 2,3-benzodiazepine type. Current Pharmaceutical Design, 8(10), 913–939. https://doi.org/10.2174/1381612024607081
Foldesi, T., Volk, B., & Milen, M. (2018). A review of 2,3-benzodiazepine-related compounds: Diazepines and 1,2,5-triazepines fused with five-membered nitrogen heterocycles. Current Organic Synthesis, 15(6), 729–754. https://doi.org/10.2174/1570179415666180601101856
Raffa, R. B., & Amantea, D. (Eds.). (2021). Naturally occurring benzodiazepines, endozepines, and their receptors: Implications for benzodiazepine therapy and withdrawal. CRC Press.
Verma, S., & Kumar, S. (2017). A mini review on synthetic approaches and biological activities of benzodiazepines. Mini-Reviews in Organic Chemistry, 14(6), 453–468. https://doi.org/10.2174/1570193X14666170511121927
Arora, N., Dhiman, P., Kumar, S., Singh, G., & Monga, V. (2020). Recent advances in synthesis and medicinal chemistry of benzodiazepines. Bioorganic Chemistry, 97, 103668. https://doi.org/10.1016/j.bioorg.2020.103668
Al-Zamani, F., & Doustkhah, E. (2022). Benzodiazepine-based drug discovery. In Heterocyclic drug discovery (pp. 1–338). Elsevier.
Gill, R. K., Kaushik, S. O., Chugh, J., Bansal, S., Shah, A., & Bariwal, J. (2014). Recent development in [1,4]benzodiazepines as potent anticancer agents: A review. Mini Reviews in Medicinal Chemistry, 14(3), 229–256. https://doi.org/10.2174/13895575113139990081
Chouinard, G. (2004). Issues in the clinical use of benzodiazepines: Potency, withdrawal, and rebound. Journal of Clinical Psychiatry, 65, 7–12.
Crocetti, L., & Guerrini, G. (2020). GABAA receptor subtype modulators in medicinal chemistry: An updated patent review (2014–present). Expert Opinion on Therapeutic Patents, 30(6), 409–432. https://doi.org/10.1080/13543776.2020.1746764
Saari, T. I., Uusi-Oukari, M., Ahonen, J., & Olkkola, K. T. (2011). Enhancement of GABAergic activity: Neuropharmacological effects of benzodiazepines and therapeutic use in anesthesiology. Pharmacological Reviews, 63(1), 243–267. https://doi.org/10.1124/pr.110.002717
Al-Kuraishy, H. M., Al-Gareeb, A. I., Saad, H. M., & El-Saber, G. B. (2023). Benzodiazepines in Alzheimer’s disease: Beneficial or detrimental effects. Inflammopharmacology, 31, 221–230. https://doi.org/10.1007/s10787-022-01099-4
Teli, S., Teli, P., Soni, S., Sahiba, N., & Agarwal, S. (2023). Synthetic aspects of 1,4- and 1,5-benzodiazepines using o-phenylenediamine: A study of past quinquennial. RSC Advances, 13, 3694–3714. https://doi.org/10.1039/D2RA06045K
Loudni, L., Roche, J., Potiron, V., Clarhaut, J., Bachmann, C., Gesson, J.-P., & Tranoy-Opalinski, I. (2007). Design, synthesis and biological evaluation of 1,4-benzodiazepine-2,5-dione-based HDAC inhibitors. Bioorganic & Medicinal Chemistry Letters, 17(17), 4819–4823. https://doi.org/10.1016/j.bmcl.2007.06.067
Mohapatra, D. K., Maity, P. K., Shabab, M., & Khan, M. I. (2009). Click chemistry based rapid one-pot synthesis and evaluation for protease inhibition of new tetracyclic triazole fused benzodiazepine derivatives. Bioorganic & Medicinal Chemistry Letters, 19(17), 5241–5245. https://doi.org/10.1016/j.bmcl.2009.06.107
Sharma, U. K., Sharma, N., Vachhani, D. D., & Van der Eycken, E. V. (2015). Metal-mediated post-Ugi transformations for the construction of diverse heterocyclic scaffolds. Chemical Society Reviews, 44(7), 1836–1860. https://doi.org/10.1039/C4CS00253A
De Silva, R. A., Santra, S., & Andreana, P. R. (2008). A tandem one-pot, microwave-assisted synthesis of regiochemically differentiated 1,2,4,5-tetrahydro-1,4-benzodiazepin-3-ones. Organic Letters, 10(20), 4541–4544. https://doi.org/10.1021/ol801841m
Yang, J., Che, X., Dang, Q., Wei, Z., Gao, S., & Bai, X. (2005). Synthesis of tricyclic 4-chloro-pyrimido[4,5-b][1,4]benzodiazepines. Organic Letters, 7(8), 1541–1543. https://doi.org/10.1021/ol050181f
Bhardwaj, P., & Kaur, N. (2023). Synthesis of 1,4-benzodiazepines by palladium-catalyzed C–N coupling. Current Organic Chemistry, 27, 282–296. https://doi.org/10.2174/1385272827666230412080929
Sergeieva, T. Y., Voskoboynik, O. Y., Okovytyy, S. I., Kovalenko, S. I., Shishkina, S. V., & Leszczynski, J. (2014). Hydrazinolysis of 3-R-[1,2,4]triazino[2,3-c]quinazolin-2-ones: Synthetic and theoretical aspects. Journal of Physical Chemistry A, 118(10), 1895–1905. https://doi.org/10.1021/jp4052616
Voskoboynik, O. Y. (2015). Synthesis, physicochemical properties and anticancer activity of 6-(heterocyclyl-N-ylmethyl)-3-R1-9-R2-2H-[1,2,4]triazino[2,3-c]quinazolin-2-ones. Voprosy Khimii i Khimicheskoi Tekhnologii, 1(99), 7–12.
Dolomanov, O. V., Bourhis, L. J., Gildea, R. J., Howard, J.A.K., & Puschmann, H. (2009). OLEX2: A complete structure solution, refinement and analysis program. Journal of Applied Crystallography, 42, 339–341. https://doi.org/10.1107/S0021889808042726
Sheldrick, G. M. (2015). SHELXT – Integrated space-group and crystal-structure determination. Acta Crystallographica Section A: Foundations and Advances, 71, 3–8. https://doi.org/10.1107/S2053273314026370
Sheldrick, G. M. (2015). Crystal structure refinement with SHELXL. Acta Crystallographica Section C: Structural Chemistry, 71, 3–8. https://doi.org/10.1107/S2053229614024218
Breitmaier, E. (2002). Structure elucidation by NMR in organic chemistry: A practical guide (3rd ed.). Wiley.
Downloads
Published
Issue
Section
License
Copyright (c) 2026 Oles Honchar Dnipro National University

This work is licensed under a Creative Commons Attribution 4.0 International License.
- Authors reserve the right of attribution for the submitted manuscript, while transferring to the Journal the right to publish the article under the Creative Commons Attribution License. This license allows free distribution of the published work under the condition of proper attribution of the original authors and the initial publication source (i.e. the Journal)
- Authors have the right to enter into separate agreements for additional non-exclusive distribution of the work in the form it was published in the Journal (such as publishing the article on the institutional website or as a part of a monograph), provided the original publication in this Journal is properly referenced
- The Journal allows and encourages online publication of the manuscripts (such as on personal web pages), even when such a manuscript is still under editorial consideration, since it allows for a productive scientific discussion and better citation dynamics (see The Effect of Open Access).

