PHENOLYSIS OF 2-(CHLOROMETHYL)OXIRANE IN THE PRESENCE OF TERTIARY AMINES: NUCLEOPHILIC-ELECTROPHILIC INTERACTIONS
DOI:
https://doi.org/10.15421/jchemtech.v34i1.352278Keywords:
epoxide ring opening, 2-(chloromethyl)oxirane (epichlorohydrin), phenolysis and acidolysis, N,N-dimethylaniline (tertiary amine), solvent polarity, regiospecificity and regioselectivity, NMR analysis, catalysis, nucleophilic-electrophilic interactions, reaction mechanismAbstract
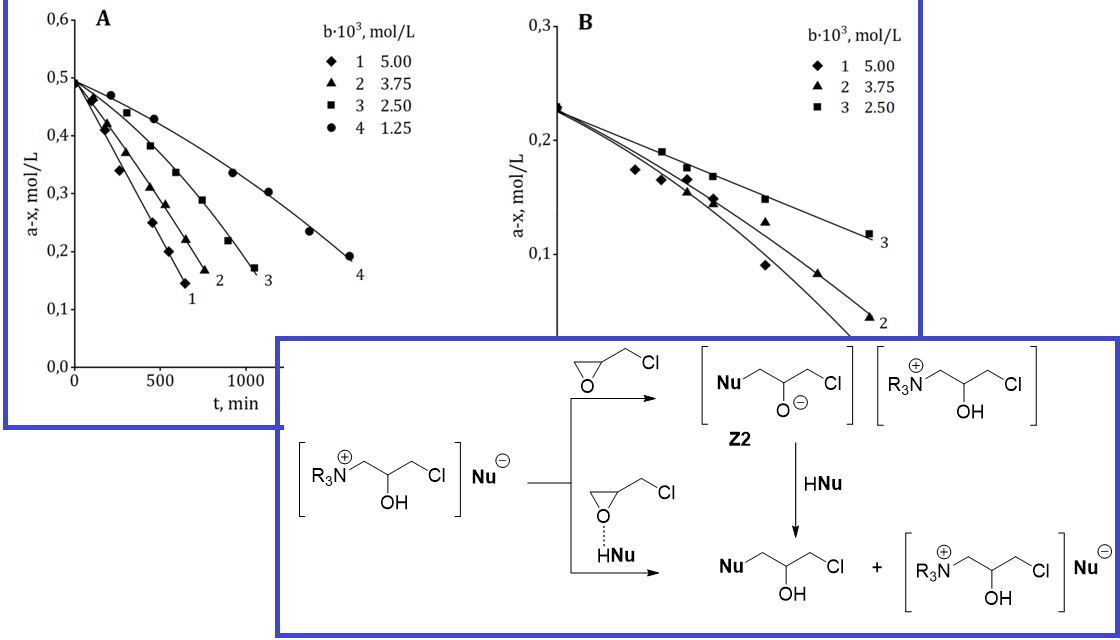
The aim of this work is to study the reaction series “4-nitrophenol–2-(chloromethyl)oxirane (epichlorohydrin, ECH)–N,N-dimethylaniline,” nucleophilic-electrophilic interactions in the system to compile a kinetic model of oxiranes phenolysis. The catalytic phenolysis of epichlorohydrin was studied using kinetic methods, along with a similar acidolysis reaction for comparison. The reaction kinetics was studied in ECH excess, which acts simultaneously as substrate and solvent, as well as in its binary mixture with tetrahydrofuran. The kinetic scheme of the reaction was confirmed by studying the structure of the synthesized product using 1H NMR and gas chromatography–mass spectroscopy. The regiospecificity of ECH phenololysis was concluded. The zero order of the reaction with respect to the nucleophilic reagent and the first order of the reaction with respect to the amine were established. The effect of solvent polarity on the reaction kinetics was examined. The kinetic law of the reaction is the same as for the catalytic acidolysis of epichlorohydrin. The kinetic features of the phenolysis and acidolysis of epichlorohydrin with varying solvent polarity were analyzed. It was confirmed that the initial stage of the reaction – amine quaternization – has an SN2 character. Nucleophilic-electrophilic interactions in the system were analyzed. The mechanism of nucleophilic opening of the oxirane cycle was described in detail. Regiospecific phenolysis and regioselective acidolysis are described by a single kinetic model that corresponds to the mechanism of transfer of the anion of a nucleophilic reagent by an ion pair.
References
Haslewood, M. N. D., Farmer, T. J., North, M. (2023). Synthesis and chemoselective crosslinking of functionalized polyesters from bio-based epoxides and cyclic anhydrides. J. Polym. Sci., 61(4), 311–322. doi.org/10.1002/pol.20220552
Muniappan Thirumalaikumar. (2022). Ring Opening Reactions of Epoxides. A Review. Organic Preparations and Procedures International, 54(1), 1–39. doi.org/10.1080/00304948.2021.1979357
Chen, J., Nie, X., Liu, Z, Mi, Z., Zhou, Y. (2015). Synthesis and application of polyepoxide cardanol glycidyl ether as biobased polyepoxide reactive diluent for epoxy resin. ACS Sustain. Chem. Eng., 3(6), 1164–1171. doi.org/10.1021/acssuschemeng.5b00095
Kasian, L. I., Kasian, A. O., Okovityi, S. I., Tarabara, I. N. (2003). [Alicyclic epoxy compounds. Reactivity]. Dnipropetrovsk, Ukraine: Izdatelstvo Dnepropetrovskogo universiteta (in Russian).
Soni, H., Verma, K., Singh, K., Priya, S. D., Bhattu, M., Singh, J. (2025). Waste derived 0D nanomaterials for the catalysis reduction of p-nitrophenol: A technological progress and developments. J. Mol. Struct., 1322(3), 140532. doi.org/10.1016/j.molstruc.2024.140532
Hanif, M., Mansha, A., Ali, K.G., Saeed, M. A., Mahmood, S., Chaudhry, A. R., Irfan, A., Mushtaq, A., Zahoor, A. F. (2025). Ring opening of epoxides: a facile approach towards the synthesis of polyketides and related stereoenriched natural products: a review. Mol. Divers., 29(5), 4919–4952. doi.org/10.1007/s11030-024-11057-7
Bespalko, Y., Sinel’nikova, M., Shved, E., Bakhalova, E. (2021). Experimental and computational studies of the mechanism of base-catalyzed ring opening of 2-(chloromethyl)oxirane by benzoic acid. Int. J. Chem. Kinet., 53(3), 356–368. doi.org/10.1002/kin.21448
Shved, E. N. (2014). [Unpublished doctoral dissertation abstract]. Ukrainian State University of Chemical Technology, Dnipropetrovsk, Ukraine. (In Ukrainian).
Potts, D. S., Locht, H., Kim, S., Lercher, J. A., Hu, J. Z., Flaherty, D. W. (2025). In Situ NMR and Kinetics Reveal Origins of Regioselectivity Differences for Epichlorohydrin Ring-Opening in Lewis and Brønsted Acid Zeolites. Angew. Chem. Int. Ed., 64 (50), e202511944. doi.org/10.1002/anie.202511944
Kumar, V., Chimni, S. S. (2023). Metal-Free Ring-Opening of Epoxides. Chemistry Select, 8(35), e202301963. doi.org/10.1002/slct.202301963
Liu, J. W., Tang, W., Wang, C. (2023). Nickel-Catalyzed Regio- and Enantioselective Ring Opening of 3,4-Epoxy Amides and Esters with Aromatic Amines. Chem. Eur. J., 29(31), e202300704. doi.org/10.1002/chem.202300704
Golsanamlou, S., Tarlani, A., Narimani, K., Hassani, E, Aghapoor, K. (2024). Regioselective ROH-Epoxystyrene-Opening over MWCNTs-[N4] Macrocycle Comprising Cu(II), Fe(III) or Cr(III). J Iran Chem Soc., 21(12), 2961–2973. doi.org/10.1007/s13738-024-03123-6
Makhin, M. N, Klebanov, M. S. (2020). Reaction kinetics of dehydrochlorination of the chlorohydrin esters of Bisphenol A. Kinet Catal., 61(2), 238–241. doi.org/10.1134/S0023158420020081
Zhang, Q., Zhou, Y. B., Chen, Y., Zhao, J. P. (2021). Ethoxylation of Phenols Catalyzed by Metal-Free Lewis Pairs: Living/Controlled Polymerization in a Slow-Initiation Mode†. Chin. J. Chem., 39 (9), 2579–2587. doi.org/10.1002/cjoc.202100289
Gansäuer, A. (2021). From Enantioselective to Regiodivergent Epoxide Opening and Radical Arylation - Useful or Just Interesting? Synlett, 32(05), 447–456. doi.org/10.1055/s-0040-1706407
Hansen, T., Vermeeren, P., Haim, A., van Dorp, M. J. H, Codée, J. D. C., Bickelhaupt, F. M., Hamlin, T. A. (2020). Regioselectivity of Epoxide Ring-Openings via SN2 Reactions Under Basic and Acidic Conditions. European Journal of Organic Chemistry. 25, 3822-3828. doi.org/10.1002/ejoc.202000590.
Yutilova, K., Tkach, A., Tarasenko, V., Shved, E. (2024). Finding the Right Balance between Tertiary Amine Steric Effect and Solvent Polarity for the Regioselectivity and Kinetics of Epichlorohydrin Acetolysis. J. Phys. Org. Chem., 37(1), e4573. doi.org/10.1002/poc.4573
Bakhtin, S., Shved, E., Bespalko, Y., Tyurina, T., Palchykov, V. (2020). Detailing the elementary stages in the oxirane ring opening reactions with carboxylic acids catalyzed by tertiary amines. J. Phys. Org. Chem., 33, 4071. doi.org/10.1002/poc.4071
Liu, F. H., Wu, X. M., Chen, K., Yao, J. B., Ran, Q. C. (2025). The Effect of Tertiary Amines as Catalysts on the Ring-Opening Polymerization of Benzoxazines. Polymers, 17(11), 1431. doi.org/10.3390/polym17111431
Bakhalova, Y. A. (2023). Benzoic acids as the proton-donating nucleophiles in reaction with 2-(chloromethyl)oxirane in the presence of bases [Doctoral thesis, Vasyl’ Stus Donetsk National University]. (In Ukrainian).
Bakhtin, S., Shved, E., Bespal’ko, Y., Stepanova, Y. (2019). Behavior modelling of organic bases in the oxyalkylation reaction of Proton-Containing nucleophiles. Prog. React. Kinet. Mech., 43 (2), 121–135. doi.org/10.3184/146867818X15161889114501
Yan, Z., Ma, Z., Deng, J., Luo, G. (2021). Mechanism and Kinetics of Epoxide Ring-Opening with Carboxylic Acids Catalyzed by the Corresponding Carboxylates. Chemical Engineering Science, 242, 116746. doi.org/10.1016/j.ces.2021.116746
Yutilova, K., Sokolovskyi, D., Shved, E., Gorden, A. E. V. (2026). Steric and Electronic Control of Regioselectivity in Salophen Metal Complex-Catalyzed Epichlorohydrin Acetolysis. J. Org. Chem., 91 (1), 163–176. doi.org/10.1021/acs.joc.5c02193
Lide, D. R. (2004). Ed. CRC Handbook of Chemistry and Physics. CRC Press.
Yutilova, K., Shved, E., & Chervonchenko, I. (2022). Sposib oderzhannia 2-(khlormetyl)oksyranu [Method of obtaining 2-(chloromethyl)oxirane] (Ukraine Patent No. 152096). Vasyl’ Stus Donetsk National University.
Yutilova, K. S., Martseniuk, N. S., Shved, E. N. (2023). Quantum Chemicai Study of the Steric Effect of Substituent on the Reactivity of Tertiary Amines in the Reaction with 2-(Chloromethyl)oxirane. Journal of Chemistry and Technologies, 31(3), 451–459. doi.org/10.15421/jchemtech.v31i3.283788
Yutilova, K. S., Bakhalova, E. A., Shved, E. N., Kravchuk, A.V., Lisova, L. S. (2023). Nucleophilic Epoxide Ring Opening in the System “Epichlorohydrin–Carboxylic Acids–Tetrabutylamonium Iodine–Solvent (ɛ=15.1–28.7). Vopr. Khimii i Khimicheskoi Tekhnologii, 2, 159–167. doi.org/10.32434/0321-4095-2023-147-2-159-167
Reichardt, C. T., Welton, T. (2011). Solvents and Solvent Effects in Organic Chemistry. Fourth. Weinheim: Wiley-VCH.
Armarego, W. L. F., Chai, C. (2013). Purification of Laboratory Chemicals. Waltham: Elsevier.
Downloads
Published
Issue
Section
License
Copyright (c) 2026 Oles Honchar Dnipro National University

This work is licensed under a Creative Commons Attribution 4.0 International License.
- Authors reserve the right of attribution for the submitted manuscript, while transferring to the Journal the right to publish the article under the Creative Commons Attribution License. This license allows free distribution of the published work under the condition of proper attribution of the original authors and the initial publication source (i.e. the Journal)
- Authors have the right to enter into separate agreements for additional non-exclusive distribution of the work in the form it was published in the Journal (such as publishing the article on the institutional website or as a part of a monograph), provided the original publication in this Journal is properly referenced
- The Journal allows and encourages online publication of the manuscripts (such as on personal web pages), even when such a manuscript is still under editorial consideration, since it allows for a productive scientific discussion and better citation dynamics (see The Effect of Open Access).

