ELECTROCHEMICAL SYNTHESIS AND SPECTROPHOTOMETRIC DETERMINATION OF MALEINATE COMPLEXES OF Cu+
DOI:
https://doi.org/10.15421/jchemtech.v32i1.294751Keywords:
Cu maleate complexes, quantum chemical modelling, electrochemical synthesis, spectrophotometryAbstract
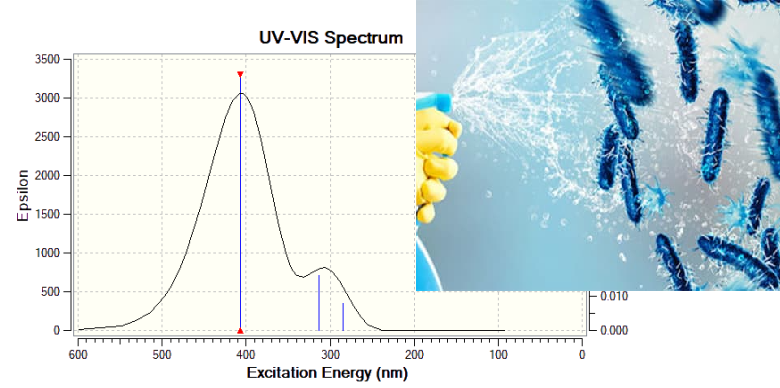
The study explored the process of anodic ionization of copper in an aqueous solution of maleic acid (H2M): we substantiated the optimal conditions for the electrochemical synthesis of Cu+ maleate complexes, ensuring nearly a 100 % yield of the target product under the following parameters: potentiostatic mode, anode potential of 0.1 V (vs. silver/silver chloride electrode), concentration of (H2M) = 0.1 M (pH = 2), and temperature (t°) = 60 °C. By utilizing spectrophotometry, along with quantum-chemical modeling of absorption spectra for various complex structures, we determined the composition of the obtained product as [Cu+(H2O)3(HM-)]. We chose the diagnostic criterion for identifying the nature of the complex as the difference in wavelength absorption for light in the π-bond of Cu+ with the sp2-hybridized carbon atom of the vinyl fragment of the maleate ion (around 400 nm) and the σ-bond of Cu+ with the ionized carboxyl group (around 300 nm). For the quantitative analysis of the working solution regarding the content of maleate π-complexes of Cu+, we suggest using the A, C-dependence measured at 360 nm.
References
Qi, Y.Y., Gan, Q., Liu, Y.X., Xiong, Y.H., Mao, Z.W., Le, X.Y. (2018). Two new Cu (II) dipeptide complexes based on 5-methyl-2-(2′-pyridyl) benzimidazole as potential antimicrobial and anticancer drugs: Special exploration of their possible anticancer mechanism. European Journal of Medicinal Chemistr; 154, 220–232. https://doi.org/10.1016/j.ejmech.2018.05.023
Shoair, A. F., El-Bindary, A. A., El-Ghamaz, N. A., Rezk, G. N. (2018). Synthesis, characterization, DNA binding and antitumor activities of Cu (II) complexes. Journal of Molecular Liquids, 269, 619–638. https://doi.org/10.1016/j.molliq.2018.08.075
Qi J., Yao Q., Tian L., Wang Y., (2018). Piperidylthiosemicarbazones Cu (II) complexes with a high anticancer activity by catalyzing hydrogen peroxide to degrade DNA and promote apoptosis. European Journal of Medicinal Chemistry, 158, 853–862. https://doi.org/10.1016/j.ejmech.2018.09.034
Kasare, M. S., Dhavan, P. P., Jadhav, B. L., Pawar, S. D. (2019). In-vitro antibacterial activity of Ni (II), Cu (II), and Zn (II) complexes incorporating new azo-azomethine ligand possessing excellent antioxidant, anti-inflammatory activity and protective effect of free radicals against plasmid DNA. Synthetic Communications, 49(23), 3311–3323. https://doi.org/10.1080/00397911.2019.1663213
Azam, M, Al-Resayes, S. I., Wabaidur, S. M., Altaf, M., Chaurasia, B. (2018). Synthesis, structural characterization and antimicrobial activity of Cu (II) and Fe (III) complexes incorporating azo-azomethine ligand. Molecules, 223(4), 813. https://doi.org/10.3390/molecules23040813
Balakrishnan, S., Duraisamy, S., Kasi, M, Kandasamy, S., Sarkar, R. (2019) Syntheses, physicochemical characterization, antibacterial studies on potassium morpholine dithiocarbamate nickel (II), copper (II) metal complexes and their ligands. Heliyon, 5(5), e01687.https://doi.org/10.1016/j.heliyon.2019.e01687
Nazirkar, B., Mandewale, M., Yamgar, R. (2019). Synthesis, characterization and antibacterial activity of Cu (II) and Zn (II) complexes of 5-aminobenzofuran-2-carboxylate Schiff base ligands. Journal of Taibah University for Science, 13(1), 440–449. https://doi.org/10.1080/16583655.2019.1592316
Justina, M.I., Olukemi, B.A., Sunday, O. (2018). Study on “Synthesis, Characterization of Nanosized Copper (II) Complex and its Antimicrobial Activities”. ChemXpress, 11(2), 136.
Zhang, Z., Wang, H., Wang, Q., Yan, M., Wang, H.. (2016). Anticancer activity and computational modeling of ternary copper (II) complexes with 3-indolecarboxylic acid and 1, 10-phenanthroline. International Journal of Oncology, 49(2), 691–699. https://doi.org/10.3892/ijo.2016.3542
Hussain, A., AlAjmi , M.F., Rehman, M.T., Amir, S., Husain, F.M. Alsalme A., Siduqui M.A., AlKhedhairy A.A., Khan R.A.. (2019). Copper (II) complexes as potential anticancer and Nonsteroidal antiinflammatory agents: In vitro and in vivo studies. Scientific Reports, 9(1), 1–7.
Wehbe, M., Lo, C., Leung, A.W., Dragowska, W.H., Ryan, G.M., Bally, M. B. (2017). Copper (II) complexes of bidentate ligands exhibit potent anti-cancer activity regardless of platinum sensitivity status. Investigational New Drugs, 35(6), 682–690. https://doi.org/10.1007/s10637-017-0488-2
Chakraborty, A., Dash, S.P., Panda, A.K, Acharyya, R., Biswas, A., Mukhopadhyay, S., Bhutia, S. K., Crochet, A. (2015). Synthesis, X-ray structure and in vitro cytotoxicity studies of Cu (I/II) complexes of thiosemicarbazone: special emphasis on their interactions with DNA. Dalton Transactions, 44(13), 6140–6157. https://doi.org/10.1039/C4DT03764B
Balakrishna, M.S., Suresh, D., Rai, A., Mague, J.T., Panda, D. (2010). Dinuclear copper (I) complexes containing cyclodiphosphazane derivatives and pyridyl ligands: synthesis, structural studies, and antiproliferative activity toward human cervical and breast cancer cells. Inorganic Chemistry, 49(19), 8790–8801. https://doi.org/10.1021/ic100944d
El-Boraey, H. A. (2012) Coordination behavior of tetraaza [N4] ligand towards Co (II), Ni (II), Cu(II), Cu (I) and Pd (II) complexes: synthesis, spectroscopic characterization and anticancer activity. Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy, 97, 255–262. https://doi.org/10.1016/j.saa.2012.05.077
González-Ballesteros N, Pérez-Álvarez D, Rodríguez-Argüelles M.C, Henriques M.S, Paixão J.A, Prado-López, S. (2016). Synthesis, spectral characterization and X-ray crystallographic study of new copper (I) complexes. Antitumor activity in colon cancer. Polyhedron, 119, 112–119. https://doi.org/10.1016/j.poly.2016.08.023
Vargaluyk, V. F., Polonskyy, V. A., Sklyar T.V., Stets N.V., Lahuta, O. V. (2023). Physici-chemical and bactericidal properties of copper-containing composites based on maleinate complexes Cu+. Journal of Chemistry and Technologies, 31(2), 208–215. http://dx.doi.org/10.15421/jchemtech.v31i2.275070
Slyvka, Yu. (2014). Template synthesis of Cu[4({C3H5-NHC(S)N-}2)Cl2(py)4] π-complex and its crystal construction. Visnyk Lviv University. Series Chemistry, 55(1), 115–123.
Vargalyuk, V. F., Polonskyy, V.A., Osokin, Y.S., Skok, A. Y. (2020). Influence of maleic acid on the composition and structure of organocopper dispersions obtained by chemical and electrochemical reduction of Cu2+ - ions Journal of Chemistry and Technologies, 28(3), 231–241. https://doi.org/10.15421/082025
Pavlyuk, O., Goreshnik, E. (2019). Synthesis and crystall structure of mixed halide Cu(I) complexses with 1,3-benzimdazolone. Visnyk of the Lviv University. Series Chemistry, 60(1), 170–178. https://doi.org/10.30970/vch.6001.170
Slyvka, Yu.I., Fedorchuk, A.A., Pokhodylo, N.T., Lis, T., Kityk, I.V., Mys’kiv, M.G. (2018). A Novel Copper (I) Sulfamate π-Complex Based on the 5-(Allylthio)-1-(3,5-dimethylphenyl)-1H-tetrazole Ligand: Alternating-current Electrochemical Crystalliza-tion, DFT Calculations, Structural and NLO Properties Studies. Polyhedron, 147, 86–93. https://doi.org/10.1016/j.poly.2018.03.015
Luk’yanov, М., Slyvka, Yu., Ardan, B., Mys’kiv, M. (2018). Synthesis and crystal structure of copper(I) sulfamate π-complex with 5-methyl-N-(Allyl)-1,3,4- thiadiazol-2-amine of [Cu2(C6H10N3S2)2(NH2SO3)2] composition. Visnyk of the Lviv University. Series Chemistry, 59(1), 157–163. http://dx.doi.org/10.30970/vch.5901.157
Vargalyuk, V. F., Osokin, Y. S., Polonskyy, V. A. (2021). Electrochemical synthesis of Сu+ complexes with maleic acid. Electrochemistry of today: achievements, problems and prospects a collective monograph, 120–121. http://doi:10.33609/978-966-8398-64-3.01.2021.1-191
Vargalyuk, V. F., Osokin, Y. S., Polonskyy, V. A., Glushkov, V. N. (2019). Features of (dπ-pπ)-binding of Cu(I) ions with acrylic, maleic and fumaric acids in aqueous solutions Journal of Chemistry and Technologies, 27, 148–157. http://dx.doi.org/10.15421/081916
Kurasova, Y. D., Vargaluyk, V. F., Polonskyy, V. A. (2022). Quantum chemical modeling of aquachlorocomplexes of Cu+ with acrylic, maleic and fumaric acids Journal of Chemistry and Technologies, 30(4), 530–536. http://dx.doi.org/10.15421/jchemtech.v30i4.263280
Frisch, M. J., Trucks, G. W., Schlegel, H. B., Scuseria, G. E., Robb, M. A., Cheeseman, J. R., Scalmani, G., Barone, V., Mennucci, B., Petersson, G.A., Nakatsuji, H., Caricato, M., Li, X., Hratchian, H.P., Izmaylov, A.F., Bloino, J., Zheng, G., Sonnenberg, J.L., Hada, M., Ehara, M., Toyota, K., Fukuda, R., Hasegawa, J., Ishida, M., Nakajima, T., Honda, Y., Kitao, O., Nakai, H., Vreven, T., Montgomery, J.A., Jr., Peralta, J.E., Ogliaro, F., Bearpark, M., Heyd, J.J., Brothers, E., Kudin, K.N., Staroverov, V.N., Kobayashi, R., Normand, J., Raghavachari, K., Rendell, A., Burant, J.C., Iyengar, S.S., Tomasi, J., Cossi, M., Rega, N., Millam, N.J., Klene, M., Knox, J.E., Cross, J.B., Bakken, V., Adamo, C., Jaramillo, J., Gomperts, R., Stratmann, R.E., Yazyev, O., Austin, A. J., Cammi, R., Pomelli, C., Ochterski, J.W., Martin, R.L., Morokuma, K., Zakrzewski, V.G., Voth, G.A., Salvador, P., Dannenberg, J.J., Dapprich, S., Daniels, A.D., Farkas, Ö., Foresman, J.B., Ortiz, J.V., Cioslowski, D. J. (2010). Fox Gaussian 09, Revision C. 01. Gaussian Inc.
Stewart, J. J. (2007). Optimization of parameters for semiempirical methods V: Modification of NDDO approximations and application to 70 elements. Journal of Molecular modeling, 13, 1173–1213. https://doi.org/10.1007/s00894-007-0233-4
Rezac, J., Fanfrlik, J., Salahub, D., Hobza, P. (2009). Semiempirical quantum chemical PM6 method augmented by dispersion and H-bonding correction terms reliably describes various types of noncovalent complexes. Journal of Chemical Theory and Computation, 5(7), 1749–1760. https://doi.org/10.1021/ct9000922
Vargaluyk, V. F., Polonskyy, V. A., Osokin, Y. S., Lahuta, O. V. (2021). Syntesis of copper composites containing maleic acid. Journal of Chemistry and Technologies, 29(3), 400–409. https://doi.org/10.15421/jchemtech.v29i3.241965.
Downloads
Published
Issue
Section
License
Copyright (c) 2024 Oles Honchar Dnipro National University

This work is licensed under a Creative Commons Attribution 4.0 International License.
- Authors reserve the right of attribution for the submitted manuscript, while transferring to the Journal the right to publish the article under the Creative Commons Attribution License. This license allows free distribution of the published work under the condition of proper attribution of the original authors and the initial publication source (i.e. the Journal)
- Authors have the right to enter into separate agreements for additional non-exclusive distribution of the work in the form it was published in the Journal (such as publishing the article on the institutional website or as a part of a monograph), provided the original publication in this Journal is properly referenced
- The Journal allows and encourages online publication of the manuscripts (such as on personal web pages), even when such a manuscript is still under editorial consideration, since it allows for a productive scientific discussion and better citation dynamics (see The Effect of Open Access).

